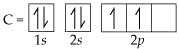
Problem 1 .
An atom has a fully occupied subshell corresponding to some orbital quantum number l. What is the number of electrons in that subshell? Explain your answer.
Problem 2 .
An atom has a fully occupied shell corresponding to some principal quantum number n. What is the number of electrons in that shell? Explain your answer.
Hint: Use the answer obtained for Problem 1 and the known mathematical formula for finite sums.
Problem 3 .
It is known that electron levels can slightly shift in energy, sometimes leading to a change in their order. For example, for a copper (Cu) atom, the state with a fully occupied 4s subshell and partially occupied 3d subshell has higher energy than the state with a fully occupied 3d subshell and partially occupied 4s subshell. Based on this, determine the electron configuration of copper. Explain your answer.
Problem 1 .
An atom has a fully occupied subshell corresponding to some orbital quantum number l. What is the number of electrons in that subshell? Explain your answer.
Problem 2 .
An atom has a fully occupied shell corresponding to some principal quantum number n. What is the number of electrons in that shell? Explain your answer.
Hint: Use the answer obtained for Problem 1 and the known mathematical formula for finite sums.
Problem 3 .
It is known that electron levels can slightly shift in energy, sometimes leading to a change in their order. For example, for a copper (Cu) atom, the state with a fully occupied 4s subshell and partially occupied 3d subshell has higher energy than the state with a fully occupied 3d subshell and partially occupied 4s subshell. Based on this, determine the electron configuration of copper. Explain your answer.