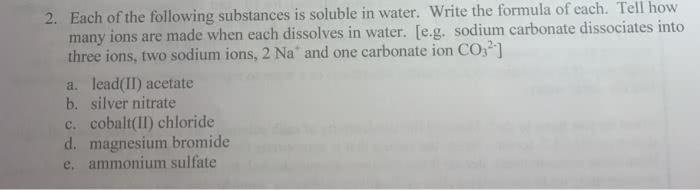1
answer
0
watching
136
views
11 Dec 2019
The following salts are soluble in water. Write the equation for the dissociation of each of the salts when added to water. One example-sodium sulfate Na2SO4(s) + aq right arrow 2Na+(ag) + SO42- (aq) A- lead (II) nitrate B- iron (II) chloride C- ammonium phosphate D- potassium carbonate 2- what is the concentration of the magnesium ions and the chloride ions in 2.5 M solution of MgCl2?
The following salts are soluble in water. Write the equation for the dissociation of each of the salts when added to water. One example-sodium sulfate Na2SO4(s) + aq right arrow 2Na+(ag) + SO42- (aq)
A- lead (II) nitrate
B- iron (II) chloride
C- ammonium phosphate
D- potassium carbonate
2- what is the concentration of the magnesium ions and the chloride ions in 2.5 M solution of MgCl2?
Keith LeannonLv2
13 Dec 2019