1
answer
0
watching
152
views
11 Dec 2019
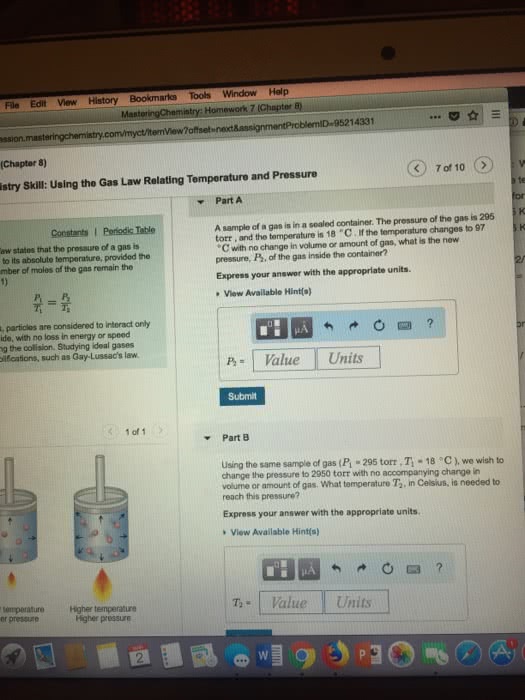
A sample of ideal gas is in a sealed container. The pressure of the gas is 235 torr , and the temperature is 37 âC . If the temperature changes to 85 âC with no change in volume or amount of gas, what is the new pressure, P2 , of the gas inside the container?
A sample of ideal gas is in a sealed container. The pressure of the gas is 235 torr , and the temperature is 37 âC . If the temperature changes to 85 âC with no change in volume or amount of gas, what is the new pressure, P2 , of the gas inside the container?
Elin HesselLv2
13 Dec 2019