1
answer
0
watching
285
views
11 Dec 2019
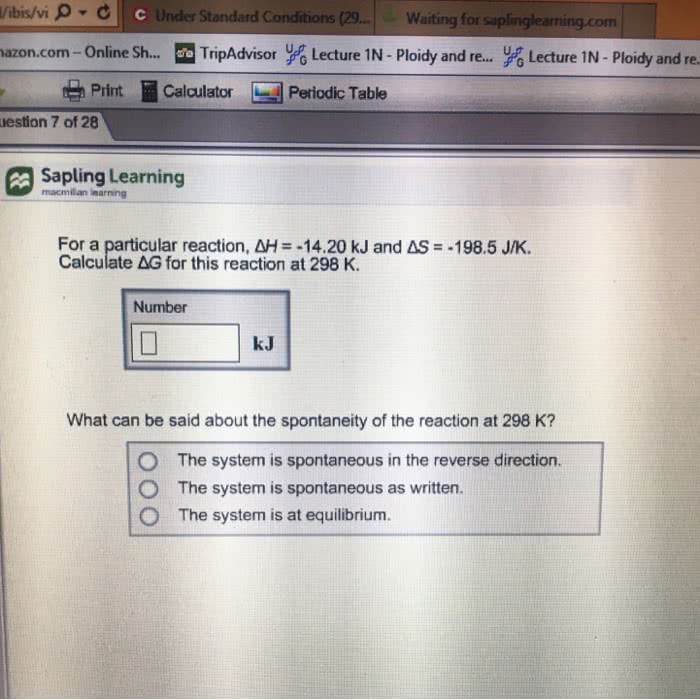
For a particular reaction, \deltaH = -144.6 kJ and \deltaS =-301.2 J/K. Calculate \deltaG for this reaction at 298 K.
Is this reaction spontaneous at 298K in the reverse direction?what about as written?
For a particular reaction, \deltaH = -144.6 kJ and \deltaS =-301.2 J/K. Calculate \deltaG for this reaction at 298 K.
Is this reaction spontaneous at 298K in the reverse direction?what about as written?
Beverley SmithLv2
13 Dec 2019