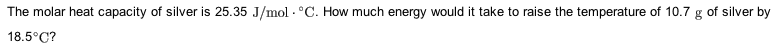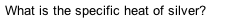0
answers
0
watching
99
views
11 Dec 2019
It takes 49.0J to raise the temperature of an 11.5g piece ofunknown metal from 13.0?C to 24.3?C. What is the specific heat forthe metal?
Express your answer numerically, in J/g??C
----------------------------------------------------------------------------------------------------
The molar heat capacity of silver is 25.35 J/mol??C. How muchenergy would it take to raise the temperature of 11.5g of silver by10.1?C?
Express your answer numerically, in joules.
-------------------------------------------------------------------------------------------------------------
What is the specific heat of silver?
It takes 49.0J to raise the temperature of an 11.5g piece ofunknown metal from 13.0?C to 24.3?C. What is the specific heat forthe metal?
Express your answer numerically, in J/g??C
----------------------------------------------------------------------------------------------------
The molar heat capacity of silver is 25.35 J/mol??C. How muchenergy would it take to raise the temperature of 11.5g of silver by10.1?C?
Express your answer numerically, in joules.
-------------------------------------------------------------------------------------------------------------
What is the specific heat of silver?