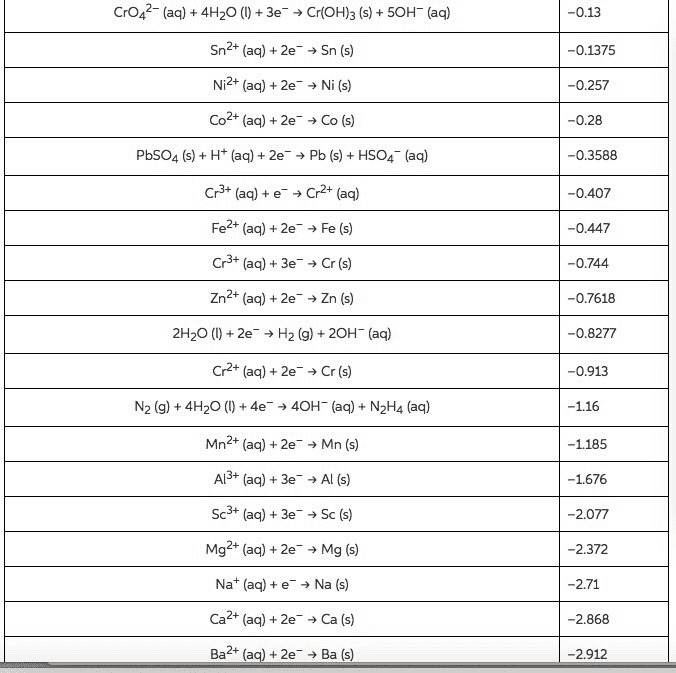
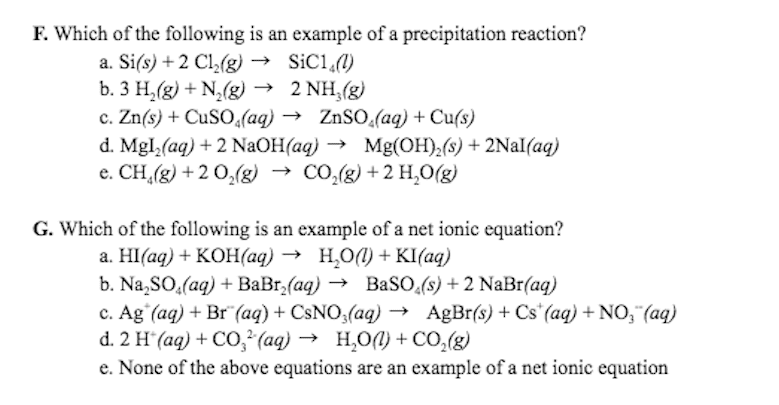2. Write a balanced chemical equation for the reaction of one anion in each of the categories in question 1.
A. a precipitation reaction
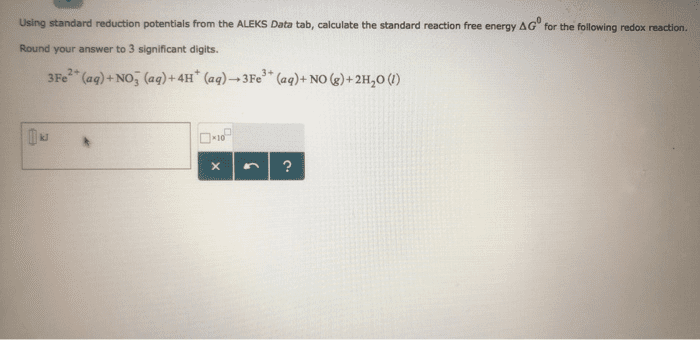
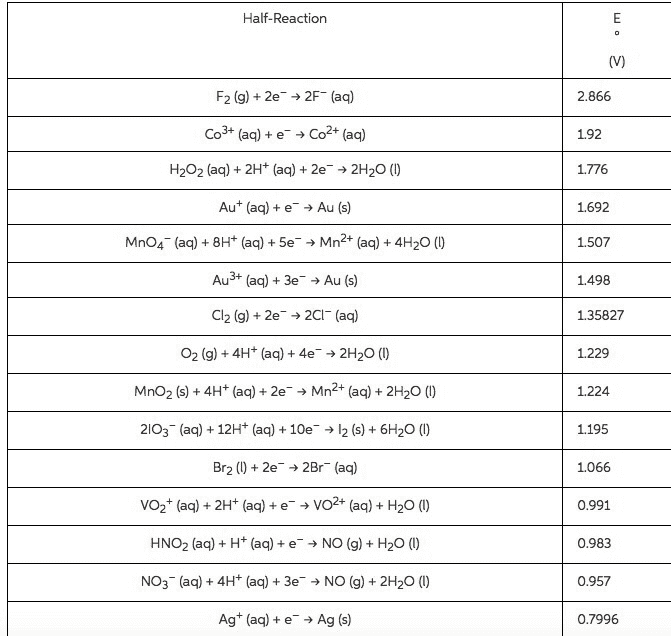
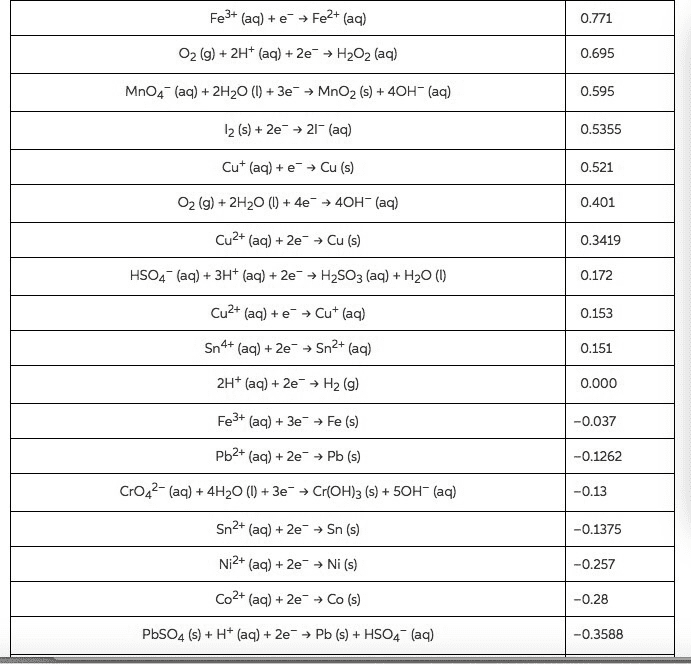
B. a redox reaction
C. an acid-base reaction
D. a gas-forming reaction
The Equations Given:
Ca+2(aq) + SO4 -2(aq) â CaSO4(s) Equation 1
Ca+2(aq) + CO3 -2(aq) â CaCO3(s) Equation 2
CaCO3(s) + 2H+ (aq) â Ca+2(aq) + CO2(g) + H2O(l) Equation 3
CaSO4(s) + H+ (aq) â No Reaction (precipitate remains) Equation 4
2NO2 - (aq) â NO(aq) + NO3 - (aq) Equation 5
2NO(g) (colorless) + O2(g) â 2NO2 (g) (red-brown) Equation 6
Ag+ (aq) + Cl- (aq) â AgCl(s) Equation 7
Ag+ (aq) + Br- (aq) â AgBr(s) Equation 8
Ag+ (aq) + I - (aq) â AgI(s) Equation 9
Cl2(aq) (colorless) + Cl- (aq) (colorless) â No Reaction Equation 10
Cl2(aq) (colorless) + Br- (aq) (colorless) â Br2(aq) (yellow to brown ) + Cl- (aq) (colorless) Equation 11
Cl2(aq) (colorless) + I - (aq) (colorless) â I2(aq) (yellow to brown) + Cl- (aq) (colorless) Equation 12
NO3 - (aq) + 4H+ (aq) + 3Fe2+(aq) â NO(aq) + 3Fe3+(aq) + 2H2O(l) Equation 13
2NO(g) (colorless) + O2(g) â 2NO2(g) (red-brown) Equation 14
2. Write a balanced chemical equation for the reaction of one anion in each of the categories in question 1.
A. a precipitation reaction
B. a redox reaction
C. an acid-base reaction
D. a gas-forming reaction
The Equations Given:
Ca+2(aq) + SO4 -2(aq) â CaSO4(s) Equation 1
Ca+2(aq) + CO3 -2(aq) â CaCO3(s) Equation 2
CaCO3(s) + 2H+ (aq) â Ca+2(aq) + CO2(g) + H2O(l) Equation 3
CaSO4(s) + H+ (aq) â No Reaction (precipitate remains) Equation 4
2NO2 - (aq) â NO(aq) + NO3 - (aq) Equation 5
2NO(g) (colorless) + O2(g) â 2NO2 (g) (red-brown) Equation 6
Ag+ (aq) + Cl- (aq) â AgCl(s) Equation 7
Ag+ (aq) + Br- (aq) â AgBr(s) Equation 8
Ag+ (aq) + I - (aq) â AgI(s) Equation 9
Cl2(aq) (colorless) + Cl- (aq) (colorless) â No Reaction Equation 10
Cl2(aq) (colorless) + Br- (aq) (colorless) â Br2(aq) (yellow to brown ) + Cl- (aq) (colorless) Equation 11
Cl2(aq) (colorless) + I - (aq) (colorless) â I2(aq) (yellow to brown) + Cl- (aq) (colorless) Equation 12
NO3 - (aq) + 4H+ (aq) + 3Fe2+(aq) â NO(aq) + 3Fe3+(aq) + 2H2O(l) Equation 13
2NO(g) (colorless) + O2(g) â 2NO2(g) (red-brown) Equation 14