1
answer
0
watching
189
views
12 Dec 2019
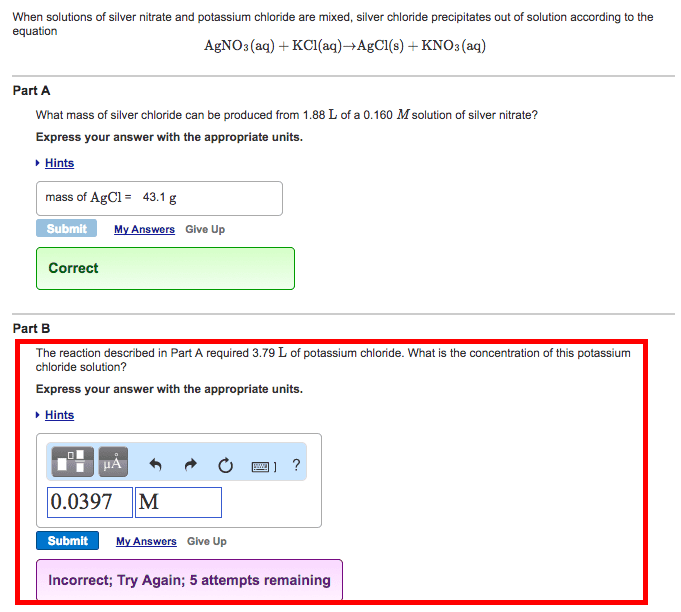
When a solution of silver nitrate is mixed with a solution of potassium chloride, a white solid precipitate forms.
Calculate the moles of solid produced when starting with a solution containing 97.80 g of silver nitrate [molar mass = 170 g/mol] (assuming that the reaction goes to completion).
Give your answer to three significant figures.
AgNO3 + KCl --> AgCl + KNO3
When a solution of silver nitrate is mixed with a solution of potassium chloride, a white solid precipitate forms.
Calculate the moles of solid produced when starting with a solution containing 97.80 g of silver nitrate [molar mass = 170 g/mol] (assuming that the reaction goes to completion).
Give your answer to three significant figures.
AgNO3 + KCl --> AgCl + KNO3
Nelly StrackeLv2
13 Dec 2019