1
answer
0
watching
1,993
views
12 Dec 2019
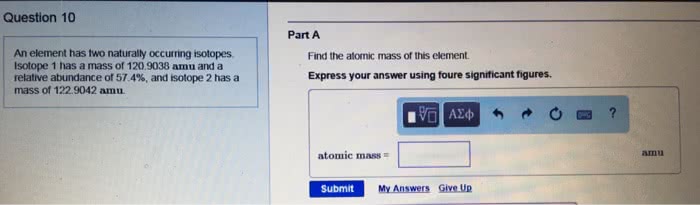
What is the atomic mass of a hypothetical element that consists of the following isotopes in the indicated natural abundances?
Express your answer to three significant figures and include the appropriate units.
Isotope Isotopic mass
(amu ) Relative abundance
(% ) 1 82.9 10.1 2 86.9 13.5 3 87.9 76.4
What is the atomic mass of a hypothetical element that consists of the following isotopes in the indicated natural abundances?
Express your answer to three significant figures and include the appropriate units.
| Isotope | Isotopic mass (amu ) | Relative abundance (% ) |
| 1 | 82.9 | 10.1 |
| 2 | 86.9 | 13.5 |
| 3 | 87.9 | 76.4 |
Nelly StrackeLv2
13 Dec 2019