1
answer
0
watching
682
views
12 Dec 2019
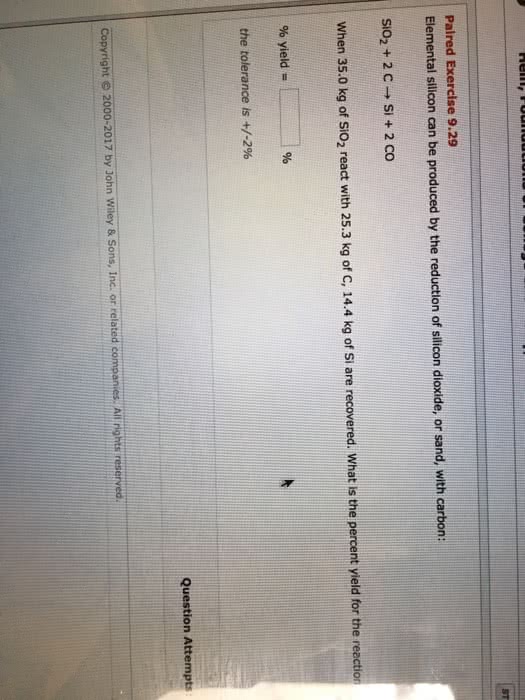
. Determine the limiting reactant when a mixture of 5.00 Ã 103 g SiO2 and 5.00 x 103 g carbon react. What is the theoretical yield of silicon carbide?
SiO2(s) + C(s) â SiC(s) + CO(g)
. Determine the limiting reactant when a mixture of 5.00 Ã 103 g SiO2 and 5.00 x 103 g carbon react. What is the theoretical yield of silicon carbide?
SiO2(s) + C(s) â SiC(s) + CO(g)
1
answer
0
watching
682
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Nelly StrackeLv2
13 Dec 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232