1
answer
0
watching
318
views
13 Dec 2019
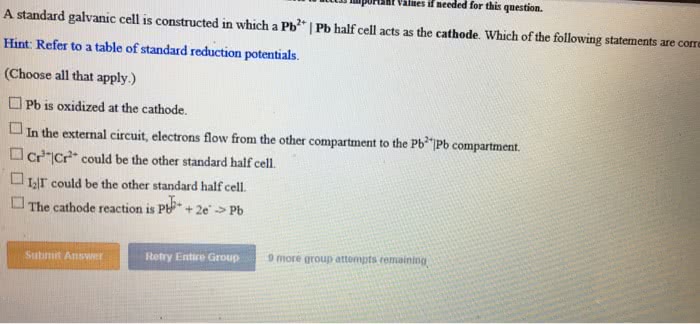
A standard galvanic cell is constructed in which a Cd2+ | Cd half cell acts as the cathode. Which of the following statements are correct?
Hint: Refer to a table of standard reduction potentials.
Cd2+ : -0.403 Fe3+ : 0.771
(Choose all that apply.)
___As the cell runs, anions will migrate from the Cd2+|Cd compartment to the other compartment.
___Cd is oxidized at the cathode.
___In the external circuit, electrons flow from the Cd2+|Cd compartment to the other compartment.
___Cd2+ is reduced at the cathode.
___The anode compartment could be Fe3+|Fe2+.
A standard galvanic cell is constructed in which a Cd2+ | Cd half cell acts as the cathode. Which of the following statements are correct?
Hint: Refer to a table of standard reduction potentials.
Cd2+ : -0.403 Fe3+ : 0.771
(Choose all that apply.)
___As the cell runs, anions will migrate from the Cd2+|Cd compartment to the other compartment.
___Cd is oxidized at the cathode.
___In the external circuit, electrons flow from the Cd2+|Cd compartment to the other compartment.
___Cd2+ is reduced at the cathode.
___The anode compartment could be Fe3+|Fe2+.
Reid WolffLv2
17 Dec 2019