1
answer
0
watching
226
views
13 Dec 2019
(i)
(a) Identify the species with the larger radius in the following pair, giving brief reason(s): Cl or S?
(b) Predict which atom has the greater first ionisation energy, giving brief reasons: P or S?
(c) Select the atom that has the greater electron affinity in the following pair, giving reasons: C or O
(d) The number of unpaired electrons in a Mn2+ ion.
(e) The number of orbitals with quantum number n=3, L=2 and ml=0
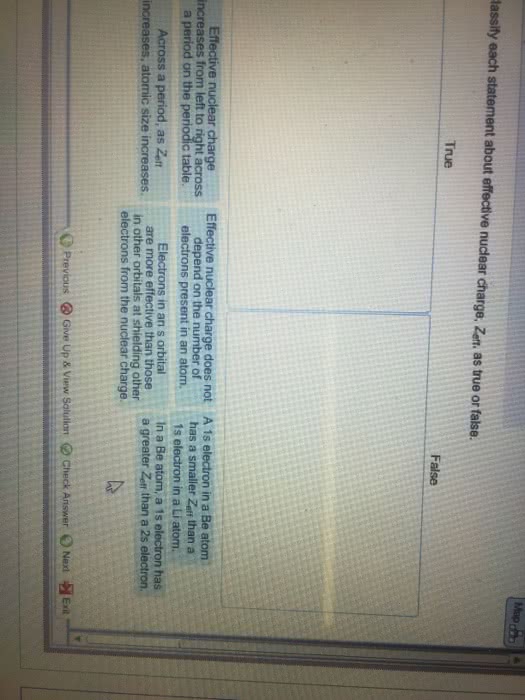
(f) Effective nuclear charge felt by a 3p and a 3d electron of a nickel atom (Z=28)
(i)
(a) Identify the species with the larger radius in the following pair, giving brief reason(s): Cl or S?
(b) Predict which atom has the greater first ionisation energy, giving brief reasons: P or S?
(c) Select the atom that has the greater electron affinity in the following pair, giving reasons: C or O
(d) The number of unpaired electrons in a Mn2+ ion.
(e) The number of orbitals with quantum number n=3, L=2 and ml=0
(f) Effective nuclear charge felt by a 3p and a 3d electron of a nickel atom (Z=28)
Elin HesselLv2
17 Dec 2019