2
answers
0
watching
266
views
29 Oct 2019
Part A
Determine whether each of the following cations is acidic or pH-neutral.
Drag the appropriate items to their respective bins.
Acidic
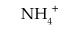
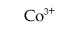
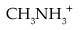
pH-neutral
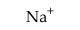
PART A IS ALREADY ANSWERED.
Part B
For NH4+, write an equation that shows how the cation acts as an acid.
Express your answer as a chemical equation. Identify all of the phases in your answer.
Part C
For Co3+, write an equation that shows how the cation acts as an acid.
Express your answer as a chemical equation. Identify all of the phases in your answer.
Part D
For CH2NH3+, write an equation that shows how the cation acts as an acid.
Express your answer as a chemical equation. Identify all of the phases in your answer.
NIH
Part A
Determine whether each of the following cations is acidic or pH-neutral.
Drag the appropriate items to their respective bins.
| Acidic
pH-neutral
|
PART A IS ALREADY ANSWERED.
Part B
For NH4+, write an equation that shows how the cation acts as an acid.
Express your answer as a chemical equation. Identify all of the phases in your answer.
| | ||
Part C
For Co3+, write an equation that shows how the cation acts as an acid.
Express your answer as a chemical equation. Identify all of the phases in your answer.
| | ||
Part D
For CH2NH3+, write an equation that shows how the cation acts as an acid.
Express your answer as a chemical equation. Identify all of the phases in your answer.
| | ||
NIH
16 Mar 2024
Nelly StrackeLv2
21 Apr 2020
Already have an account? Log in


