1
answer
0
watching
309
views
13 Dec 2019
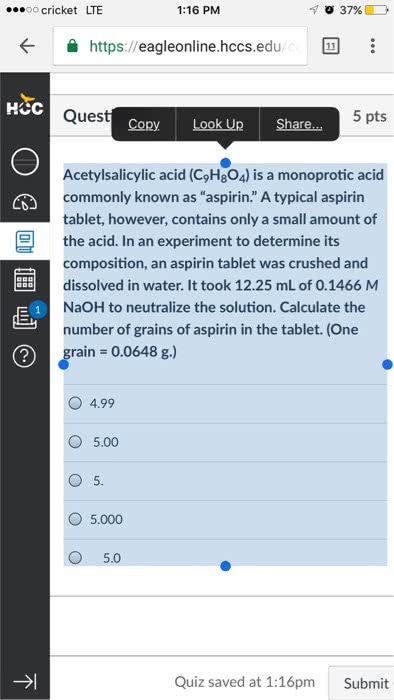
What is the pH at 25?C of the solution obtained by dissolving a5.00 grain tablet of aspirin in 0.500 liter of water. The tabletcontains 5.00 grains or 0.324 grams of aspirin, HC9H7O4. The acidis monoprotic and the Ka = 3.3 x 10^-4 at 25?C.
What is the pH at 25?C of the solution obtained by dissolving a5.00 grain tablet of aspirin in 0.500 liter of water. The tabletcontains 5.00 grains or 0.324 grams of aspirin, HC9H7O4. The acidis monoprotic and the Ka = 3.3 x 10^-4 at 25?C.
Nestor RutherfordLv2
17 Dec 2019