1
answer
0
watching
716
views
6 Oct 2020
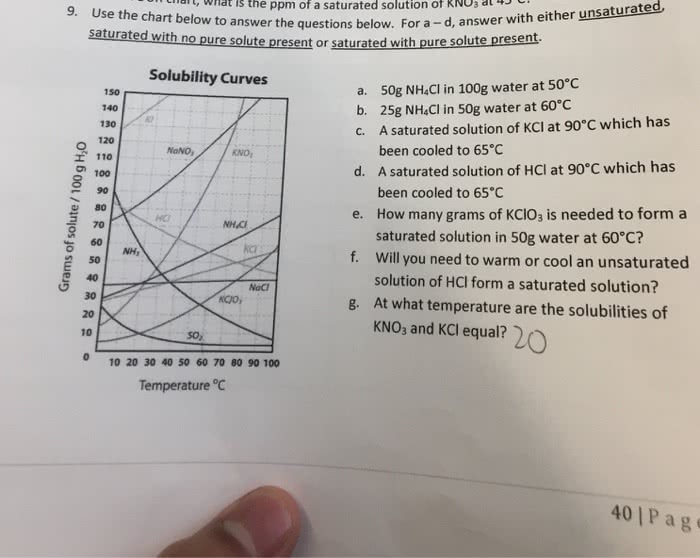
Sodium bicarbonate (baking soda) is commonly used to absorb odor. Its solubility is 9.6g/100g H2O at 30oC and 16g/100g H2O at 60oC. At 60oC, 9.2 g of baking soda is added to 46 g of water.
A. Is the resulting mixture homogeneous at 60oC? if not, how many grams of baking soda are undissolved?
B. The mixture is cooled to 30oC. How many more grams of water are needed to make a saturated solution?
Sodium bicarbonate (baking soda) is commonly used to absorb odor. Its solubility is 9.6g/100g H2O at 30oC and 16g/100g H2O at 60oC. At 60oC, 9.2 g of baking soda is added to 46 g of water.
A. Is the resulting mixture homogeneous at 60oC? if not, how many grams of baking soda are undissolved?
B. The mixture is cooled to 30oC. How many more grams of water are needed to make a saturated solution?
1
answer
0
watching
716
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Christian GarciaLv10
13 Nov 2020
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232