1
answer
0
watching
238
views
28 Nov 2020
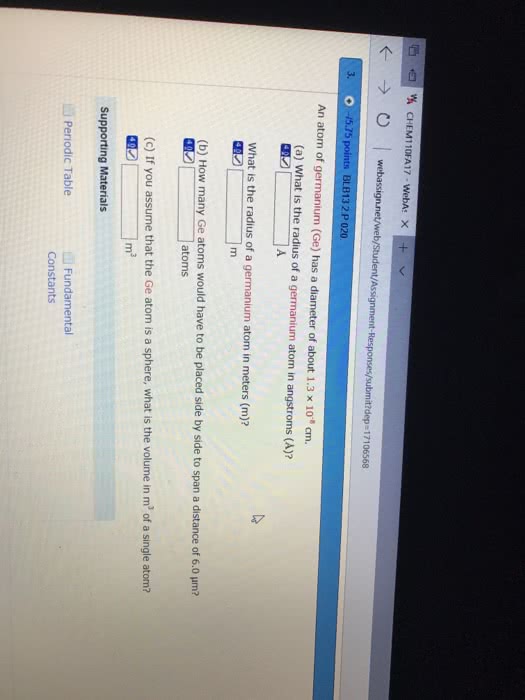
An atom of tin (

(a) What is the radius of a tin atom in angstroms (
(b) How many 

(c) If you assume that the tin atom is a sphere, what is the volume in 
An atom of tin (

(a) What is the radius of a tin atom in angstroms (
(b) How many 

(c) If you assume that the tin atom is a sphere, what is the volume in 
Miguel MatienzoLv10
30 Jan 2021