Exercise 18.44
Label the anode and cathode, indicate the direction of electron flow and the species present in each solution for each of the following overall redox reactions.
Part A
Ni2+(aq)+Mg(s)âNi(s)+Mg2+(aq)
Drag the appropriate labels to their respective targets.
Help
Reset
SubmitMy AnswersGive Up
Correct
Part B
Indicate the half-reaction occurring at Anode.
Express your answer as a chemical equation. Identify all of the phases in your answer.
SubmitMy AnswersGive Up
Part C
Indicate the half-reaction occurring at Cathode.
Express your answer as a chemical equation. Identify all of the phases in your answer.
SubmitMy AnswersGive Up
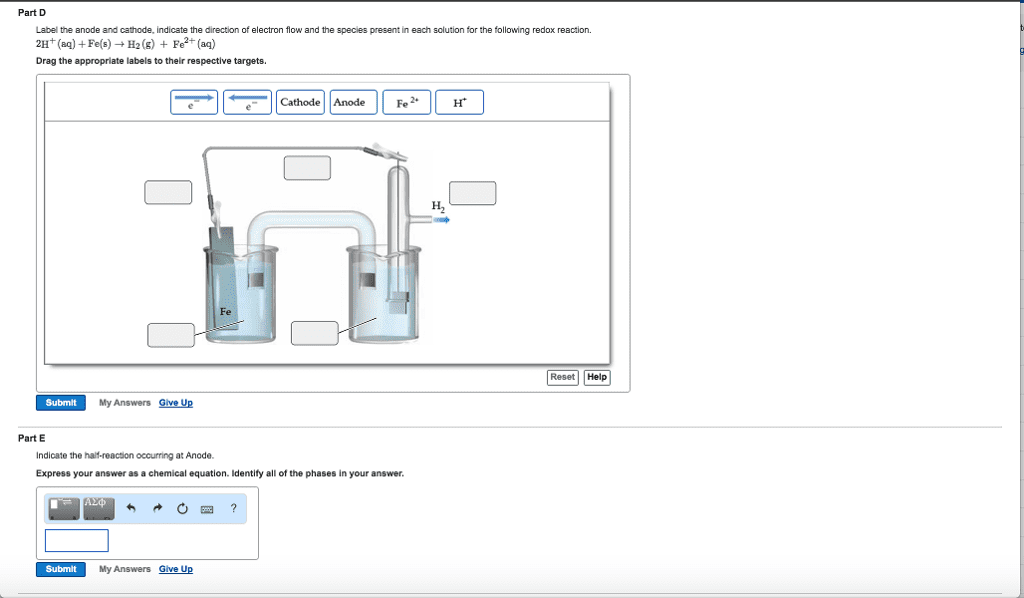
Part D
2H+(aq)+Fe(s)âH2(g)+Fe2+(aq)
Drag the appropriate labels to their respective targets.
Help
Reset
SubmitMy AnswersGive Up
Correct
Part E
Indicate the half-reaction occurring at Anode.
Express your answer as a chemical equation. Identify all of the phases in your answer.
SubmitMy AnswersGive Up
Part F
Indicate the half-reaction occurring at Cathode.
Express your answer as a chemical equation. Identify all of the phases in your answer.
SubmitMy AnswersGive Up
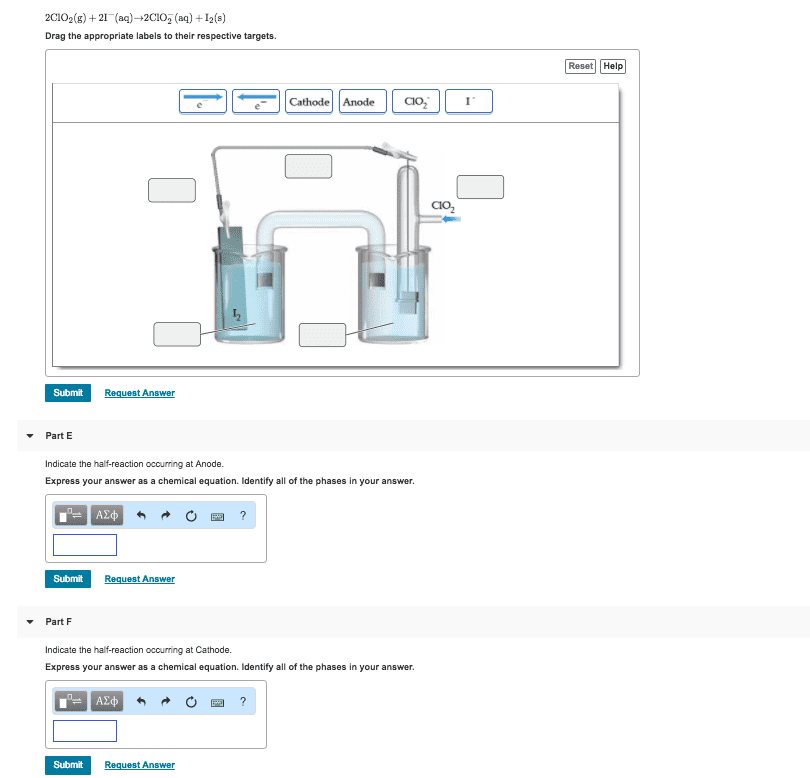
Part G
2NOâ3(aq)+8H+(aq)+3Cu(s)â2NO(g)+4H2O(l)+3Cu2+(aq)
Drag the appropriate labels to their respective targets.
Help
Reset
SubmitMy AnswersGive Up
Correct
Part H
Indicate the half-reaction occurring at Anode.
Express your answer as a chemical equation. Identify all of the phases in your answer.
SubmitMy AnswersGive Up
Part I
Indicate the half-reaction occurring at Cathode.
Express your answer as a chemical equation. Identify all of the phases in your answer.
SubmitMy AnswersGive Up
Exercise 18.44
Label the anode and cathode, indicate the direction of electron flow and the species present in each solution for each of the following overall redox reactions.
Part A
Ni2+(aq)+Mg(s)âNi(s)+Mg2+(aq)
Drag the appropriate labels to their respective targets.
| Help Reset |
SubmitMy AnswersGive Up
Correct
Part B
Indicate the half-reaction occurring at Anode.
Express your answer as a chemical equation. Identify all of the phases in your answer.
| | ||
SubmitMy AnswersGive Up
Part C
Indicate the half-reaction occurring at Cathode.
Express your answer as a chemical equation. Identify all of the phases in your answer.
| | ||
SubmitMy AnswersGive Up
Part D
2H+(aq)+Fe(s)âH2(g)+Fe2+(aq)
Drag the appropriate labels to their respective targets.
| Help Reset |
SubmitMy AnswersGive Up
Correct
Part E
Indicate the half-reaction occurring at Anode.
Express your answer as a chemical equation. Identify all of the phases in your answer.
| | ||
SubmitMy AnswersGive Up
Part F
Indicate the half-reaction occurring at Cathode.
Express your answer as a chemical equation. Identify all of the phases in your answer.
| | ||
SubmitMy AnswersGive Up
Part G
2NOâ3(aq)+8H+(aq)+3Cu(s)â2NO(g)+4H2O(l)+3Cu2+(aq)
Drag the appropriate labels to their respective targets.
| Help Reset |
SubmitMy AnswersGive Up
Correct
Part H
Indicate the half-reaction occurring at Anode.
Express your answer as a chemical equation. Identify all of the phases in your answer.
| | ||
SubmitMy AnswersGive Up
Part I
Indicate the half-reaction occurring at Cathode.
Express your answer as a chemical equation. Identify all of the phases in your answer.
| | ||
SubmitMy AnswersGive Up