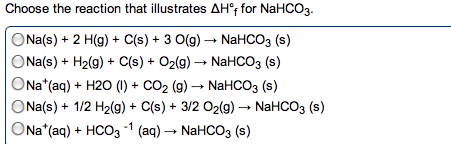
2
answers
0
watching
36
views
19 Nov 2019
Physical chemistry question

Physical chemistry question


a. Write the formation reaction for methanol, CH_3 OH (gas) b. Estimate the enthalpy of formation of methanol, CH_3 OH (liquid) using the data provided below. BE C - H = 420 BE C - O = 350 BE 0 - H = 460 kJ/mol C (s) rightarrow C (g) delta H = + 717 BE H - H = 430 BE O = O = 500 kJ/mol CH_3 OH (liquid) rightarrow CH_3 OH (gas) delta H = + 40 kJ/mol
2
answers
0
watching
36
views
For unlimited access to Homework Help, a Homework+ subscription is required.
harshit9155Lv1
10 Dec 2022
Unlock all answers
Get 1 free homework help answer.
Already have an account? Log in
Irving HeathcoteLv2
28 Sep 2019
Get unlimited access
Already have an account? Log in
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232