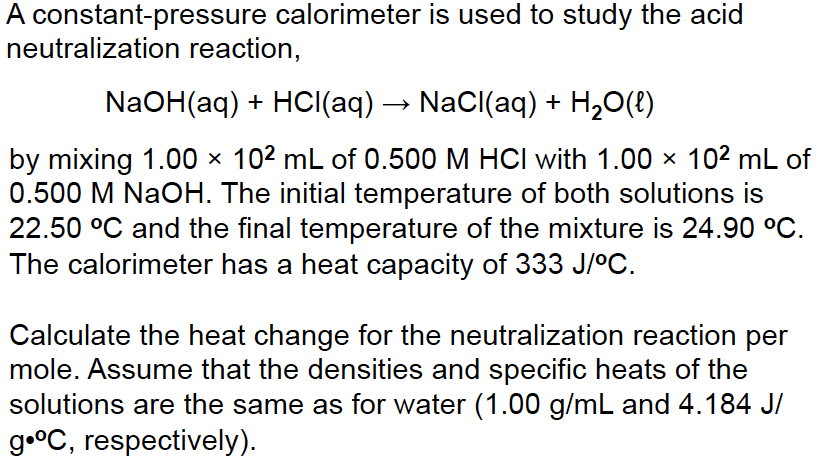2
answers
0
watching
72
views
12 Dec 2019
Given the reaction:
HCl + NaOH -> H2O + NaCl
Mass of 2.0 M HCl = 50.0 g
Mass of 2.0 M NaOH = 50.0g
Temperature of HCl before mixing = 24 celsius
Temperature of NaOH before mixing = 24 celsius
Temperature after mixing both = 37 celsius
Q solution = 2719.6 J
Q calorimeter = 679.9 J
Total heat of neutralization = -3399.5 J
Moles of acid reacted = 0.1 mole
Moles of water produced = 0.1 mole
Calculate the heat of neutralization per one mole of water formed = ____________ kJ/mole
Given the reaction:
HCl + NaOH -> H2O + NaCl
Mass of 2.0 M HCl = 50.0 g
Mass of 2.0 M NaOH = 50.0g
Temperature of HCl before mixing = 24 celsius
Temperature of NaOH before mixing = 24 celsius
Temperature after mixing both = 37 celsius
Q solution = 2719.6 J
Q calorimeter = 679.9 J
Total heat of neutralization = -3399.5 J
Moles of acid reacted = 0.1 mole
Moles of water produced = 0.1 mole
Calculate the heat of neutralization per one mole of water formed = ____________ kJ/mole
desmarcos19Lv10
16 Jan 2022
Unlock all answers
Get 1 free homework help answer.
Already have an account? Log in
Bunny GreenfelderLv2
13 Dec 2019
Get unlimited access
Already have an account? Log in