1
answer
0
watching
35
views
13 Dec 2019
Need help to find what is empty in this data/calculation page
Molarity if standard AgNO3 solution - 0.492 AgNO3 M
Mass of vial and chloride unkown - 24.1998 g
Mass of vial - 23.2177 g
Mass of unkown in volumetric flask - 0.9821 g
Mass of unknown in each 20.00-mL sample - ___g
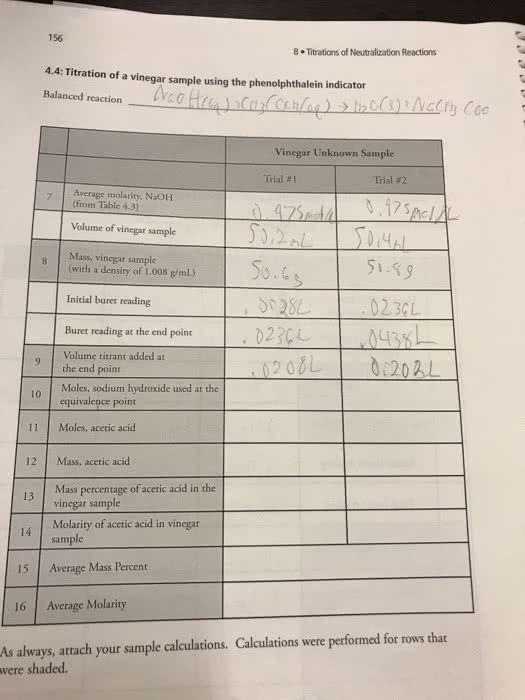
Titration I Titration II Titration III Initial Buret Reading 0.01 mL 0.01 mL 0.01 mL Final Buret Reading 38.23 mL 38.10 mL 38.70 mL Volume of AgNO3 used to titrate sample 38.22 mL 38.09 mL 38.69 mL # moles AgNO3 used to titrate sample # moles of Cl- present in sample Mass of Cl- present in sample Percentage of Cl- in sample
Average Value of Percentage Cl- in unknown: ______%
Need help to find what is empty in this data/calculation page
Molarity if standard AgNO3 solution - 0.492 AgNO3 M
Mass of vial and chloride unkown - 24.1998 g
Mass of vial - 23.2177 g
Mass of unkown in volumetric flask - 0.9821 g
Mass of unknown in each 20.00-mL sample - ___g
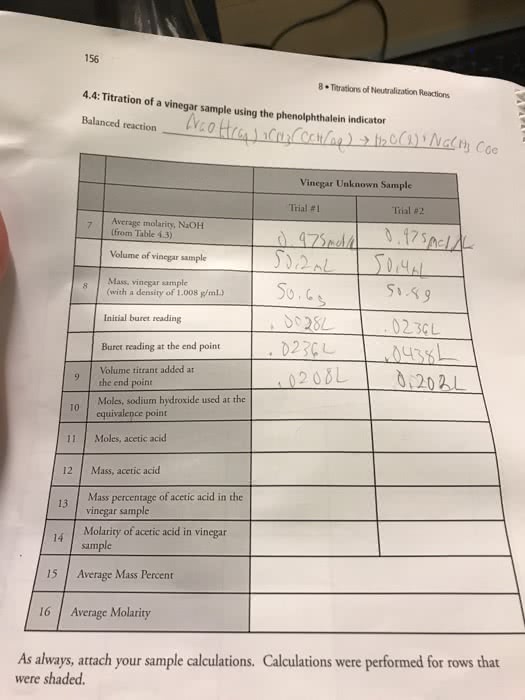
| Titration I | Titration II | Titration III | |
| Initial Buret Reading | 0.01 mL | 0.01 mL | 0.01 mL |
| Final Buret Reading | 38.23 mL | 38.10 mL | 38.70 mL |
| Volume of AgNO3 used to titrate sample | 38.22 mL | 38.09 mL | 38.69 mL |
| # moles AgNO3 used to titrate sample | |||
| # moles of Cl- present in sample | |||
| Mass of Cl- present in sample | |||
| Percentage of Cl- in sample |
Average Value of Percentage Cl- in unknown: ______%
22 Jul 2023
Unlock all answers
Get 1 free homework help answer.
Already have an account? Log in