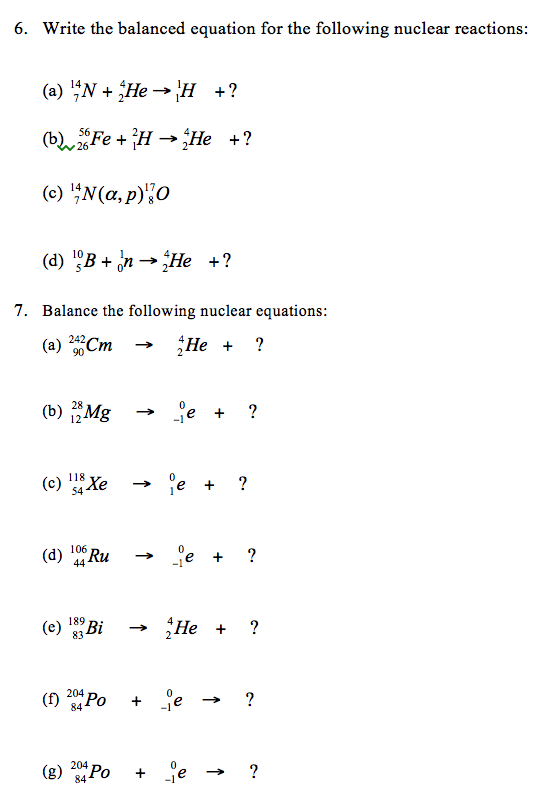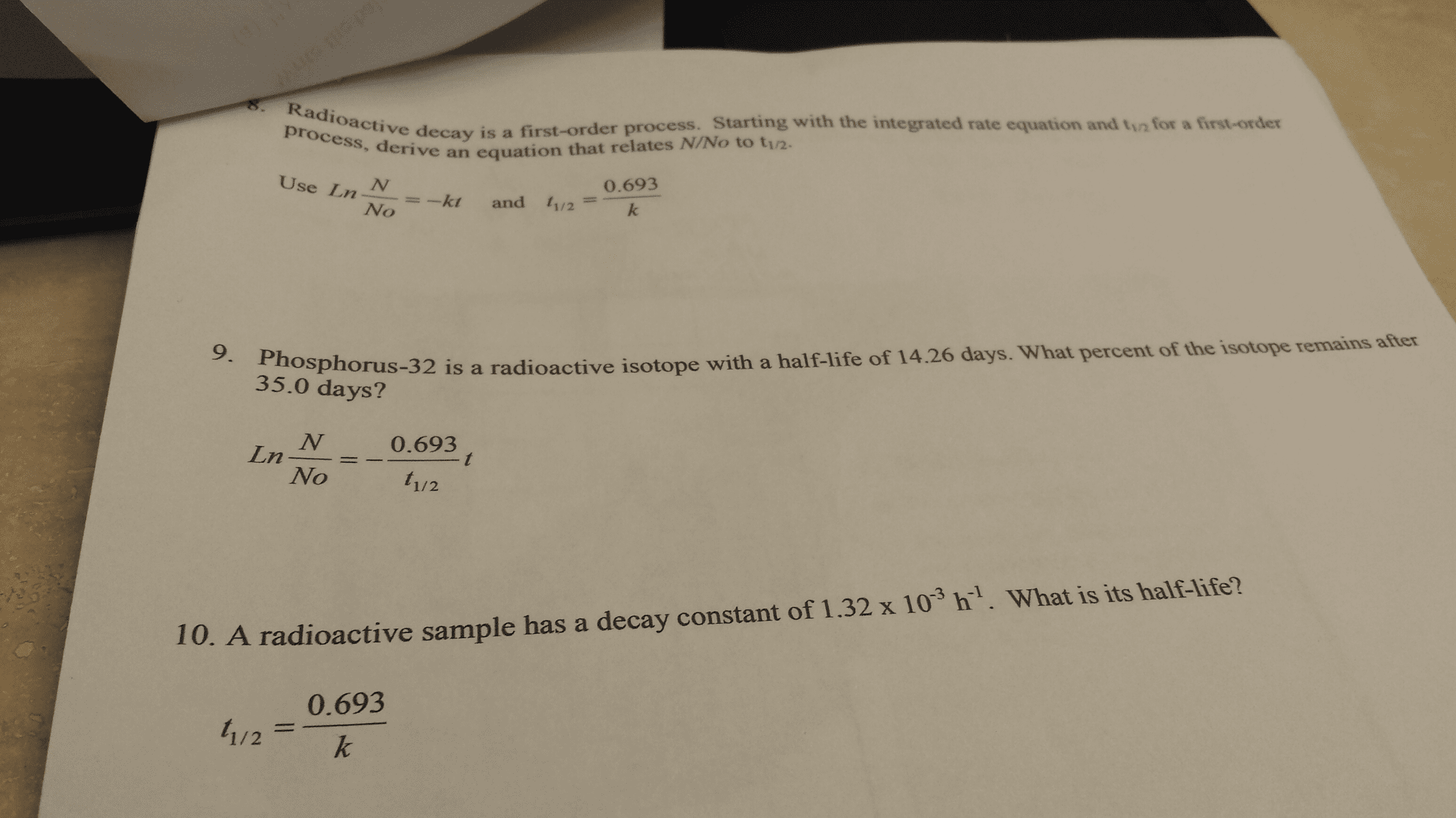Directions: Answer each of the following questions.
1. One isotope of Br has a half-life of 16.5 hours. How much of a 2.00-gram sample remains at the end of 1.00 day?
2. If 160 grams of a 200-gram sample of gold-198 decay in 6.25 days, what is the half-life of gold-198?
3. The symbol for an electron also used to represent a(n)
a. Alpha particle
b. Beta particle
c. Positron
d. Gamma-ray
4. During a fission reaction:
a. A larger and more stable nucleus is formed
b. Energy is absorbed
c. Positrons are always released
d. Smaller and more stable nuclei are formed
5. The half-life of a radioactive isotope
a. Changes over time
b. Is constant
c. Is a characteristic of stable isotopes
d. Decreases in value as time passes
6. Radioactive dating is used to determine the
a. Half-life of a radioactive isotope
b. Type of particle emitted by an isotope as it decays
c. Age of a specimen
d. Time required for half of a radioactive sample to decay
7. Carbon-11 has a half-life of 20 minutes. Explain what this means.
8. Conduct research to identify two naturally occurring radioactive elements and two synthetic radioactive elements.
CRITICAL THINKING:
9. Conduct research to explain how fusion differs from fission. How are the two processes alike? How are they different? (Compare and contrast) Don’t forget to cite your resources.
Directions: Answer each of the following questions.
1. One isotope of Br has a half-life of 16.5 hours. How much of a 2.00-gram sample remains at the end of 1.00 day?
2. If 160 grams of a 200-gram sample of gold-198 decay in 6.25 days, what is the half-life of gold-198?
3. The symbol for an electron also used to represent a(n)
a. Alpha particle
b. Beta particle
c. Positron
d. Gamma-ray
4. During a fission reaction:
a. A larger and more stable nucleus is formed
b. Energy is absorbed
c. Positrons are always released
d. Smaller and more stable nuclei are formed
5. The half-life of a radioactive isotope
a. Changes over time
b. Is constant
c. Is a characteristic of stable isotopes
d. Decreases in value as time passes
6. Radioactive dating is used to determine the
a. Half-life of a radioactive isotope
b. Type of particle emitted by an isotope as it decays
c. Age of a specimen
d. Time required for half of a radioactive sample to decay
7. Carbon-11 has a half-life of 20 minutes. Explain what this means.
8. Conduct research to identify two naturally occurring radioactive elements and two synthetic radioactive elements.
CRITICAL THINKING:
9. Conduct research to explain how fusion differs from fission. How are the two processes alike? How are they different? (Compare and contrast) Don’t forget to cite your resources.