1
answer
1
watching
154
views
rahafdubai2Lv1
18 Mar 2024
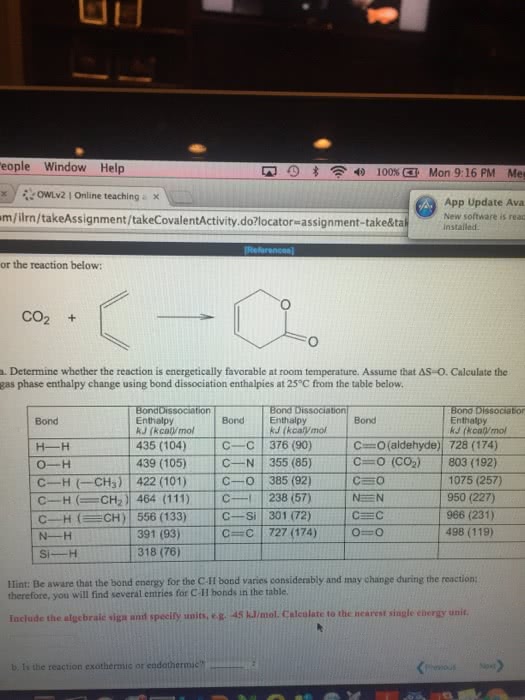
For the reaction below:

a. Estimate the gas phase enthalpy change using bond dissociation enthalpies from the OWL Table Reference, not data from your text. Click the References button and then click the Tables link on the drop-down that appears. Include algebraic sign and units.
fill in the blank 1
b. Is the reaction exothermic or endothermic?
c. Is the reaction likely to proceed spontaneously in the direction written?
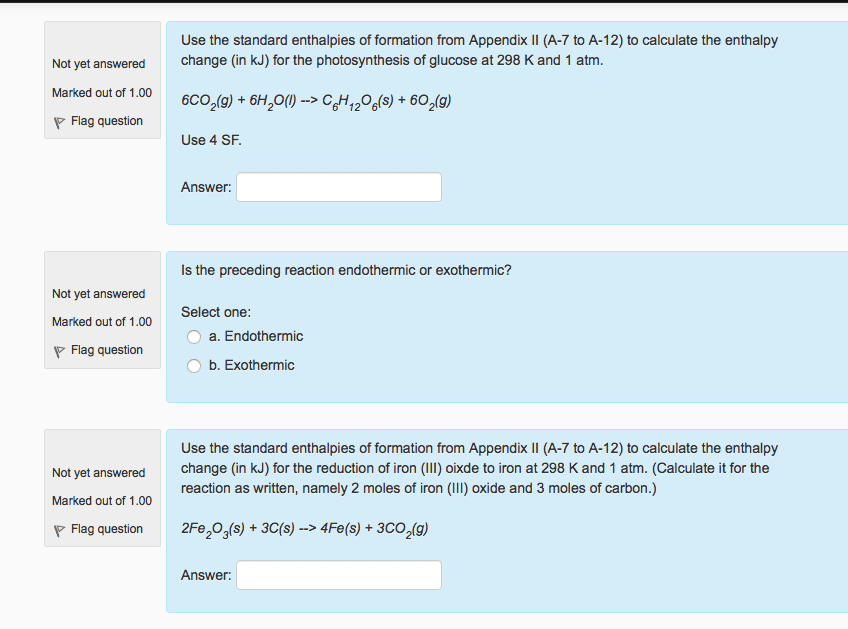
For the reaction below:
a. Estimate the gas phase enthalpy change using bond dissociation enthalpies from the OWL Table Reference, not data from your text. Click the References button and then click the Tables link on the drop-down that appears. Include algebraic sign and units.
fill in the blank 1
b. Is the reaction exothermic or endothermic?
c. Is the reaction likely to proceed spontaneously in the direction written?