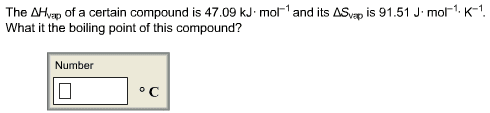CHEM 1050 Study Guide - Midterm Guide: Nitroglycerin, Reduction Potential, Permanganate
201 views15 pages
11 Jan 2017
School
Department
Course
Professor
Document Summary
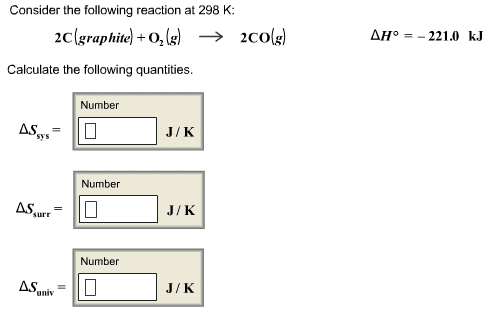
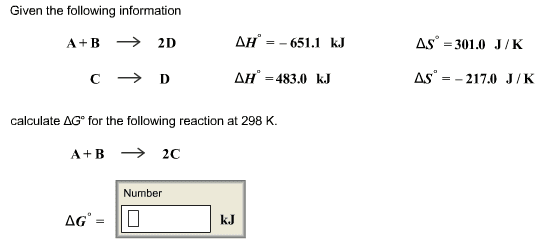
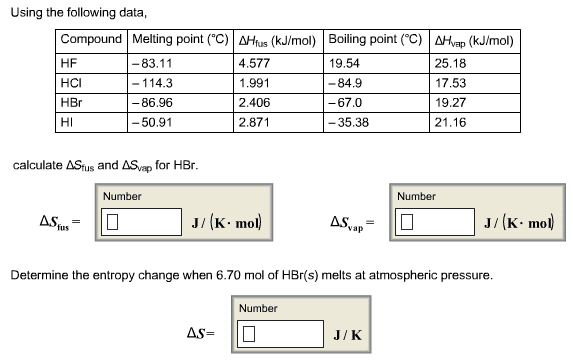
This is the beginning of midterm exam 2. Third law of entropy: s = 0 (at 0k) also, this is good to measure entropy levels of reactions. S*= [(2) (192. 5 j/k*mol)] [(1) (191. 5 j/k*mol) + (3) (130. 58 j/k*mol)] If g is negative when the reaction (forward/spontaneous reaction). G = 0 when the reaction is at equilibrium. G is positive when reaction is forward and is not spontaneous. You have h and s for making nh3. If it were not for s, all exothermic reactions would be spontaneous. S can go up or down, depending on the temperature of the reaction. Negative contribution to g increases the tendency of reaction to go as written. But if s is negative t s decreases the tendency of reaction to occur. So when h and t s are of opposite signs, relative importance of two terms will dictate if g negative or positive.
Get access
Grade+
$40 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
10 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232