In the ionic compounds LiF, NaCl, KBr, and RbI, the measured cationâanion distances are 2.01 AÌ (LiâF), 2.82 AÌ (Na-Cl), 3.30 AÌ (KâBr), and 3.67 AÌ (RbâI), respectively.
Predict the cation-anion distance of each ionic compound above using the values of ionic radii given in Figure 7.7 on page 257 in the textbook. To receive full credit, you must show how you predicted the distance for each molecule by providing a step-by-step solution for each molecule. The solution for each molecule is worth 8 points; thus, a total of 32 total points is possible for this question.
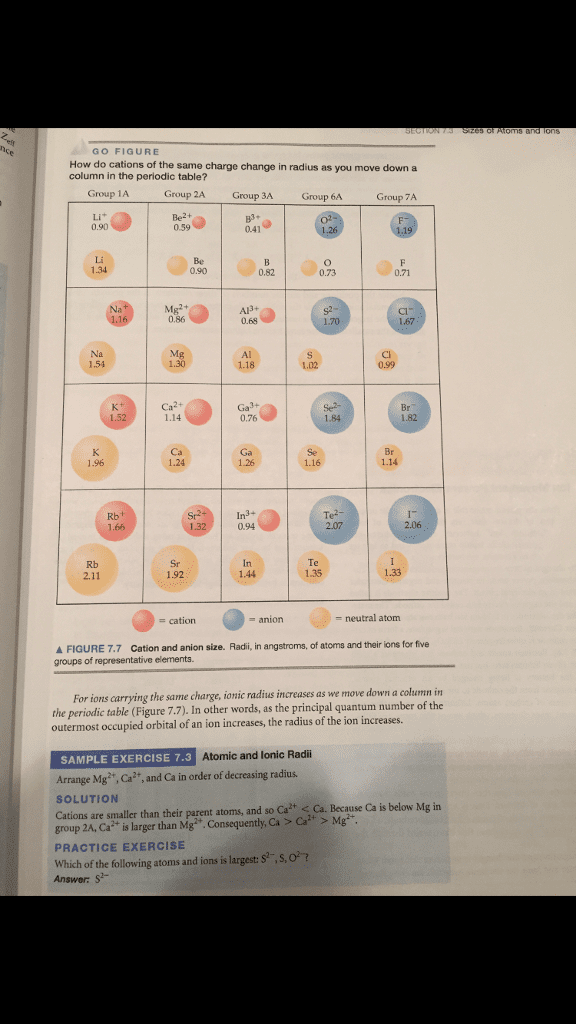
GO FIGURE How do cations of the same charge change in radius as you move down a column in the periodic table? Group IA Group 2A Group 3A Group 6A Group 7A 1.35 neutral atom cation A FIGURE 7.7 cation and anion size. Radii, in angstroms, of atoms and their ions for five groups of representative elements. For ions carrying the same charge, ionic radius increases as we move down a column in the periodic table (Figure 7.7). In other words, as the principal quantum number of the outermost occupied or of an ion increases, the radius of the ion increases. SAMPLE EXERCISE 7.3 Atomic and Ionic Radii Arrange Mg Ca2 and Ca in order of decreasing radius. SOLUTION Ca isbelow Mg in cations are than their parent atoms, and so Ca2+ Ca. Because smaller group 2A, Ca larger than Mga Ca Ca PRACTICE EXERCISE Which of the following atoms and ions is largest S2 ,s,o? Answer: s?