CHEM 1113 Study Guide - Final Guide: Matter Wave, Bohr Model, Rydberg Formula
Document Summary
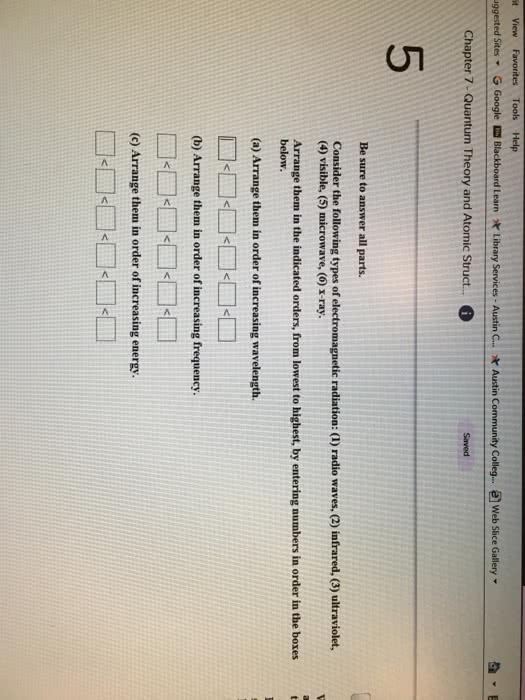
Chapter 7 - quantum theory and atomic structure concept area 1 - properties of light. Increasing wavelength-decreasing energy: gamma rays x rays ultraviolet visible infrared. Microwave radio: you should know the equation, = c, and be able to define each quantity in it, and given the wavelength of a light wave, be able to calculate its frequency and vice versa. Absorption spectrum: the spectrum produced when atoms absorb specific wavelengths of incoming light and become excited from lower to higher energy levels. Emission spectrum: the line spectrum produced when excited atoms return to lower energy levels and emit photons characteristic of the element. Graphed spectrum: x-axis is wavelength and y-axis is absorbance: you should know the equation e = h , define each quantity in the equation, and given , calculate e and vice versa. You should know the planck formula for the energy of a photon, e = h , and define each quantity in the equation.