CHM 114 Study Guide - Final Guide: Hot Rod, Steel Wool, Styrofoam
Document Summary
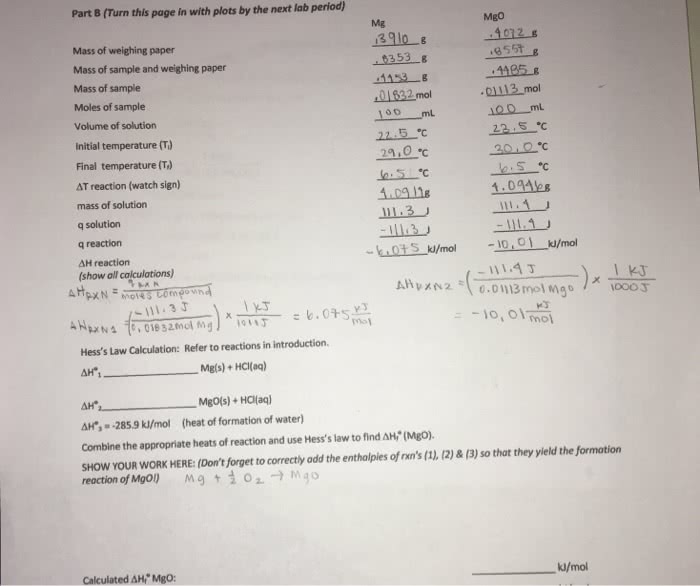
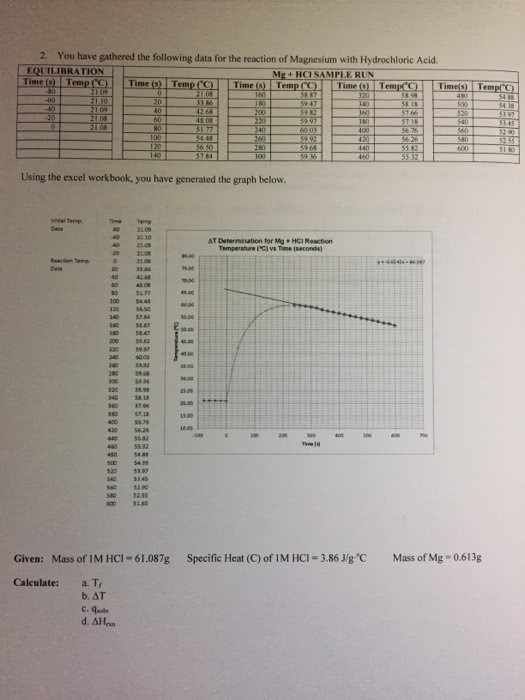
The purpose of this experiment is to determine the enthalpy of formation indirectly of mgo(s) with a colorimeter. This will be done by using a series of equations and hess"s law to form a theoretical final equation. We will also be calculating the specific heat of an unknown metal. In this experiment, we will be calculating the enthalpy of formation through hess"s law. Enthalpy of formation, also demonstrated by hf, is when one mole of a substance is formed from its constituent elements at standard states. Hess"s law tells us that the sum of all of the steps of a reaction can be added to equal the enthalpy of the final equation. This is possible because enthalpy of formation is a state function, which means it is independent of the intermediate state of the system, only the initial and final values. We will be using three equations to determine the final equation.