CHM 114 Study Guide - Final Guide: Chemical Kinetics, Cuvette, Phenolphthalein
Document Summary
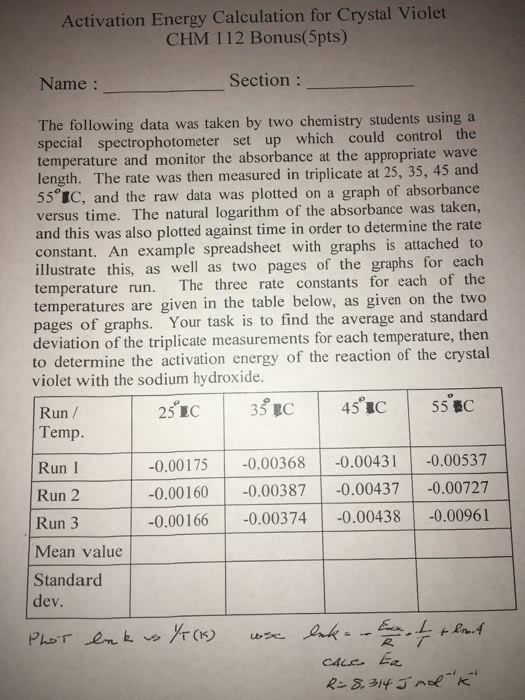
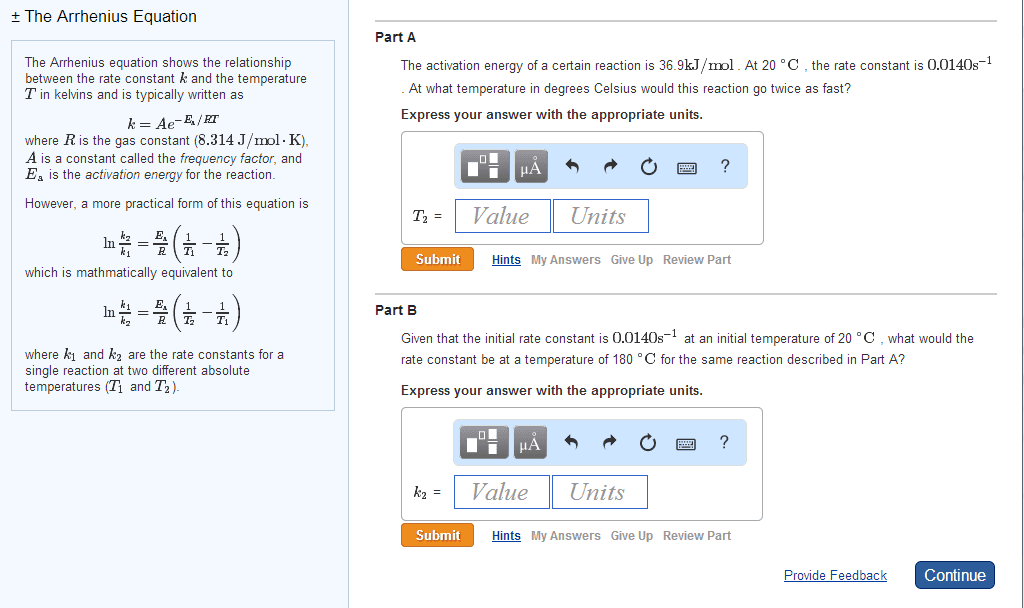
Purpose: the purpose of the experiment is to determine the activation energy of a first order reaction at room temperature and zero degrees celsius. The reaction examined is between a hydroxide ion and phenolphthalein. Theory: to find the activation energy in this experiment, we will record the absorbance of the reaction at different times. The beer- lambert law is the equation for absorbance (a=ebc) where a is the absorbance, e is the molar absorptivity, b is the length of light traveled in the cuvette, and c is the concentration of absorbing solution. Once we get the absorbance, we can see at what time the absorbance decreases to half the original absorbance. From this, we can use the half life equation to find k, which is the rate constant. When we find two different rate constants at two different temperatures, we can then use the arrhenius equation to find the activation energy.