CHEM 14A Midterm: Chem 14A Midterm 2 Study Guide
Document Summary
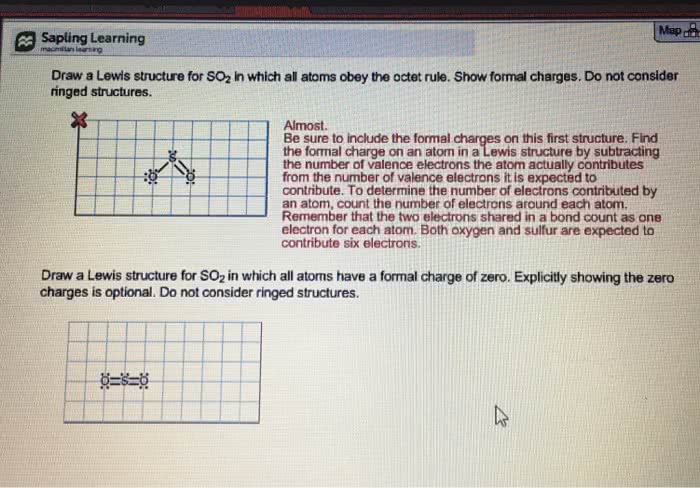
Octet rule: in a covalent bond formation, atoms go as far as possible toward completing their octets (noble-gas con guration) by sharing electron pairs. Lewis structure: a diagram shows the atoms of a molecule by their chemical symbols, covalent bonds by lines and lone pairs by pairs of dots. Writing the lewis structure: count the valence electrons and just the number for charges on ions, arrange the atoms, connect the atoms with bonding electron pairs, complete the octets, represent the bonds. Resonance: way of describing delocalized electrons within certain molecules or molecular ions where the bonding cannot be expressed by one single lewis structure; the blending of all structures. Electrons that are shown in different positions in a set of resonance structures are said to be delocalized. Delocalization: shared electron pair is distributed over several pairs of atoms and cannot be identi ed with just one pair of atoms.