Phospholipids contain a glycerol backbone that is attached totwo fatty acid chains through an ester linkage and to a phosphateion through a phosphate ester linkage. An ester linkage is formedby the condensation reaction of the hydroxyl group (?OH) of theglycerol and the carboxylic acid functional group(O || ?C?OH)of a fatty acid. A phosphate ester linkage is formed from thehydroxyl group (?OH) and the phosphate ion. The phosphate ion isthen linked to a polar organic molecule through a phosphate esterlinkage. A phospholipid molecule containing two fatty acid chainsof 12 carbon atoms each having nocarbon-carbon double bonds in thechain and a polar organic group, X, is:
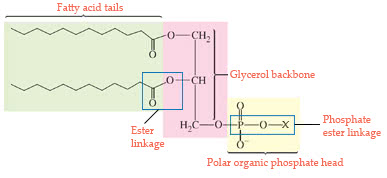
The phospholipid molecule is amphipathic, as it has a polar endand a nonpolar end.
Part A
A certain phospholipid molecule contains two fatty acid chainsof eight carbon atoms each having no carbon-carbon doublebonds in the chain. The phosphate ion is attached to a polarorganic molecule, X, just as highlighted in yellow in the figure inthe introduction.
Complete the structure of the phospholipid molecule using thestructure in the introduction. To include a X group in yourstructure, draw another atom, hover over the atom with the mouse,and then press the X key on your keyboard.




