CHEM 1301 Chapter Notes - Chapter 6.2: Oxidation State, Electronegativity, Formal Charge
Document Summary
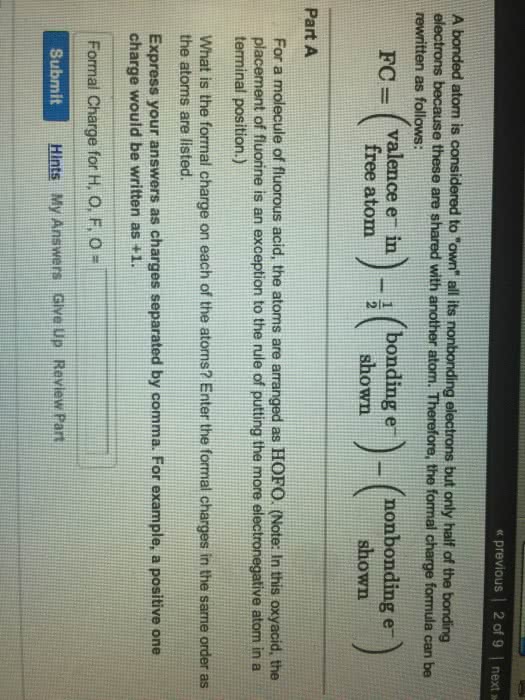
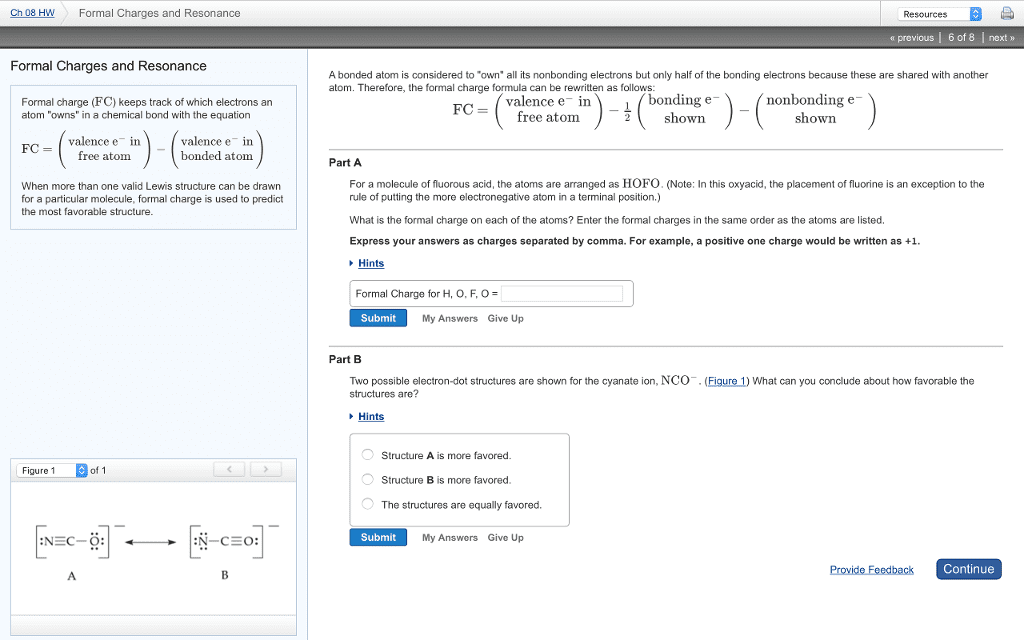
In order to determine the most plausible lewis structure, when more than one possibility exists for a compound, a book-keeping method is used, namely formal charge. The number of valence electrons distributed around each atom in lewis structure can be compared with the number of valence electrons in the free atom. The smaller these differences for molecules structure, the lower is its energy. Formal charge is a measure of the redistribution of the electrons. We ca(cid:374) (cid:373)ake a(cid:374) assu(cid:373)ptio(cid:374) that a(cid:374) ato(cid:373) (cid:862)o(cid:449)(cid:374)s(cid:863) all its lo(cid:374)e pair electro(cid:374)s a(cid:374)d half of each shared bonding pairs. Fc = v l 1/2 b. V = the number of valence electrons in the free atom. B = the number of bonding electrons in the atom. A quick way to determine the formal charge. L = the number of electrons present as lone pairs in a bonded atom: fc = number of valence electrons number of dots number of single lines around an atom.