CHEM 1301 Chapter Notes - Chapter 14.1-14.6, 16.4 : Dynamic Equilibrium, Equilibrium Constant, Elementary Reaction
42 views7 pages
2 Apr 2016
School
Department
Course
Professor
Document Summary
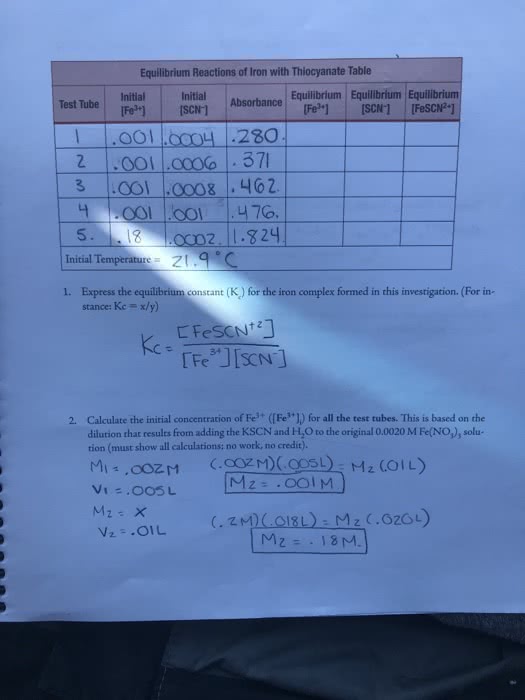
At the same ime a reacion is consuming reactants, it is also breaking up products: when the amount of products is low, the decomposiion reacion will slow down, and the composiion reacion will speed up. Eventually the rate of decomposiion will equal the rate of composiion and the rate of one reacion exactly balances the other, known as dynamic equilibrium. At dynamic equilibrium the rate of the forward and reverse reacions are equal to each other: dynamic meaning individual molecules react coninuously, equilibrium meaning, there is no net change in the system. If the forward reacion went to compleion, there would be no reactants, if the reverse reacion went to compleion there would be no product, instead we have equal amounts of both. Each the forward and reverse reacions have their own keq. At equilibrium the rates equal each other so we can make. The concentraions you arrange are products over reactants, and we don"t include solids or gases.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232