CHE-1102 Chapter Notes - Chapter 14: Chemical Kinetics, Kinetic Energy, Reaction Rate
Document Summary
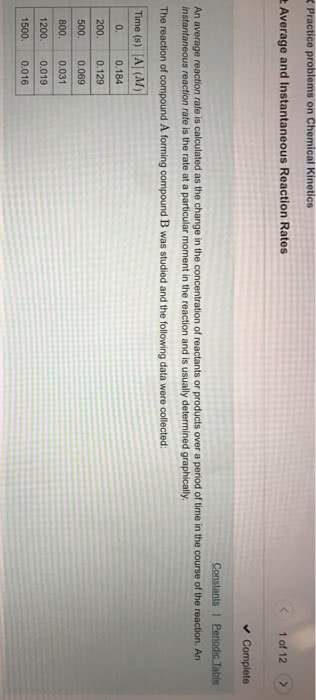
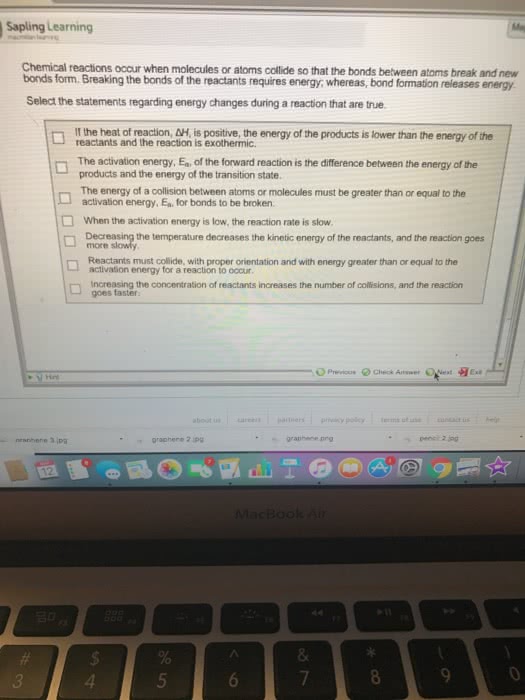
Chemical kinetics is the study of how reactions change over time. Rates of reactions are dependent on the reaction concentration, temperature, presence of catalysts, and the physical state of reactants. The probability of reactants colliding with each other increases as concentration increases. As kinetic energy increases, the chance of reactants colliding with each other also increases. Homogeneous mixtures have faster reaction rates than heterogeneous mixtures. Solids have the slowest reactions and gases have the fastest reactions. Watching the change in concentration of either the reactants or products over time can tell us the rate of the reaction. Reaction rate is the change in the concentration over the change in time. Average rate is measured over a period of time. Instantaneous rate is measured at a specific time. Reactants disappear and products appear (a b) The average rate of disappearance of a is = change in concentration of a.