CHEM 11200 Chapter Notes - Chapter 1: Ideal Gas Law, Supersaturation, Ideal Gas
129 views8 pages
23 Jun 2016
School
Department
Course
Professor
Document Summary
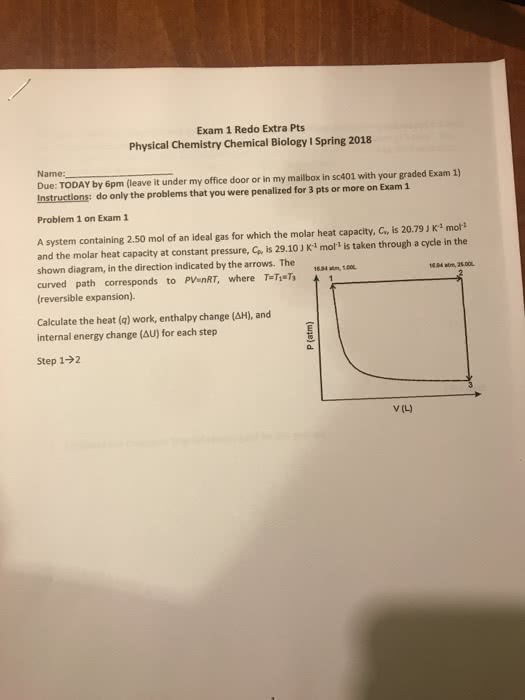
Chemistry 12200 solutions to problem set 1 due fri. jan 10, 2014 in lecture. Feel free to use these solutions to prepare the correction to turn. In on mon (to your ta"s box on the second floor) for the remaining 10 points, But of course explain why your original solution was in error. Definition of pv work done on the system (note: the work done by the system is w) E = q + w (or euniv = 0) (note: the mcats still use the old convention that e = q w so their w is the work done on the system and would be defined as ! H = qp careful, this one can only be used for const pinternal processes or reactions. Ideal gas law pv = nrt and c p = c v +r (any ideal gas)
Get access
Grade+
$40 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
10 Verified Answers
Class+
$30 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
7 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232
Related Documents
Related Questions
0.9 mol of an ideal gas initially at 19 âC and 1.15 bar pressure are heated to a final temperature of 330 âC at constant pressure. The Cv,m of this gas is 3.3R.
Part A - Calculate the initial volume of the gas.
Express your answer in m3 .
| |
| Vi = |
| m3 |
SubmitMy AnswersGive Up
Part B - Calculate the final volume of the gas.
Express your answer in m3 .
| |
| Vf = |
| m3 |
SubmitMy AnswersGive Up
Part C - Calculate the work done on/by the gas.
Express your answer in kJ. Pay attention to the sign!
| |
| w = |
| kJ |
SubmitMy AnswersGive Up
Part D - Calculate the change in internal energy.
Express your answer in kJ. Pay attention to the sign!
| |
| ÎU = |
| kJ |
SubmitMy AnswersGive Up
Part E - Calculate the heat absorbed/released by the gas.
Express your answer in kJ. Be careful with signs!
| |
| q = |
| kJ |