CHEM 101 Chapter Notes - Chapter 2: Exothermic Process, Unified Atomic Mass Unit, Intensive And Extensive Properties
Document Summary
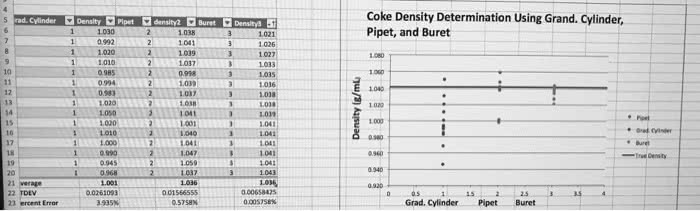
Accuracy- a term that refers to how close a measured value is to the actual value. Precision- a term that refers to how close a series of measurements are to one another or how reproducible they are. Random error- error that has equal probability of being too high or too low. Systemaic error- error that tends toward being consistently either too high or too low. Measurements usually involve the use of instruments, which have an inherent amount of uncertainty. In reported measurements, every digit is certain except the last, which is esimated. The precision of a measurement refers to its reproducibility. The accuracy of a measurement refers to how close a measurement is to the actual value of the quanity being measure d )- the raio of an object"s mass to its volume. Intensive property- a property such as density that is independent of the amount of a given substance.