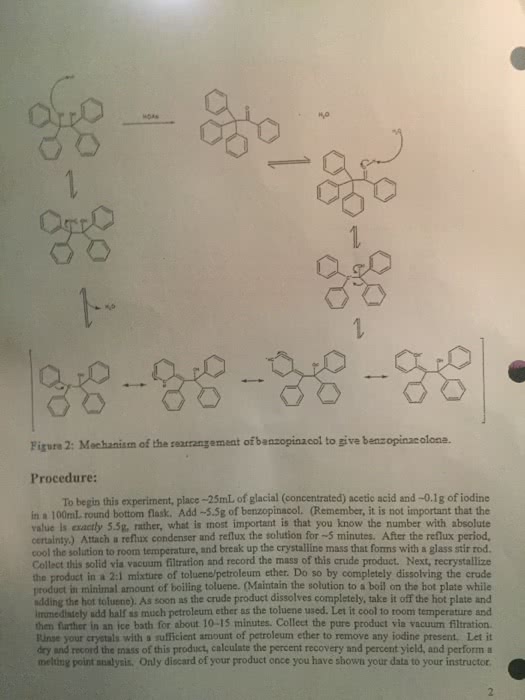
CHEM 211 Chapter Notes - Chapter 7.8-7.10: Methyl Group, Alkyne, Carbocation
Document Summary
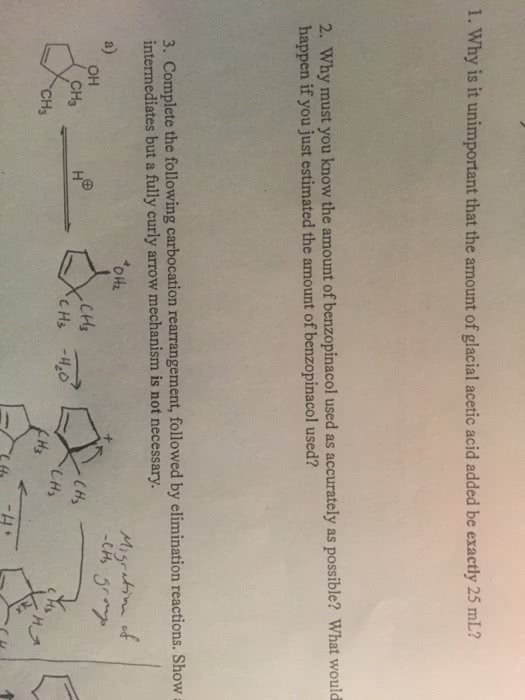
Rearrangement occurs when a less stable, secondary carbocation rearranges to a more stable tertiary carbocation: this occurs when a methyl group migrates from the carbon atoms adjacent to the charged carbon. It migrates as a ch3-, making the positive carbon a neutral one, and the old carbon the adjacent carbon was attached to, a positively charged one. This kind of rearrangement is called a 1,2 shift: the transition state for rearrangement, the methyl group never leaves the molecule, it just forms partially broken and partially formed bonds. The major and minor product is dictated by which of the products is more stable of an alkene: zaitsev"s rule. Other kinds of rearrangements: hydrogens can also migrate to form more stable carbocations (=hydride migration, the migration of a methyl group is called methanide migration, ring size can also change based off rearrangement activity. Very likely to occur if it relieves ring strain.