CHEM 24112 Chapter Notes - Chapter 7: Barium Chloride, Ammonium Sulfate, Potassium Sulfate

Homework 7.2: Net Ionic Equations and Acid-Base Reactions
1. What is a net ionic equation? What species are shown in such an equation, and which species are
not shown?
2. What are spectator ions? Write an example of an equation in which spectator ions are present and
identify them.
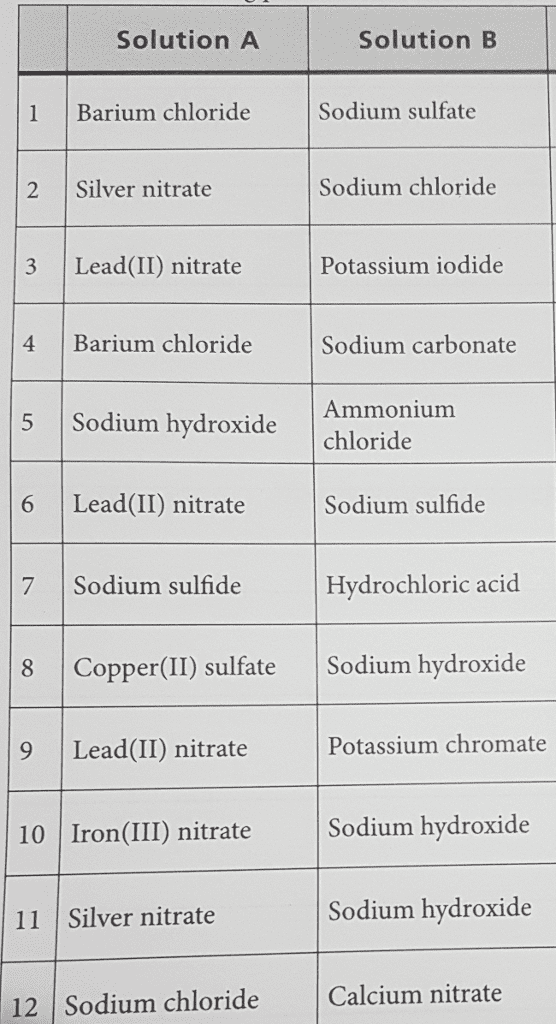
3. Write balanced net ionic equations for the reactions that occur when the following aqueous
solutions are mixed. If no reaction is likely (i.e. there is no driving force), so indicate.
a. silver nitrate, AgNO3, and potassium chloride, KCl
b. nickel (II) sulfate, NiSO4, and barium chloride, BaCl2
c. ammonium phosphate, (NH4)3PO4, and calcium chloride, CaCl2
d. hydrofluoric acid, HF, and potassium sulfate, K2SO4
e. calcium chloride, CaCl2, and ammonium sulfate, (NH4)2SO4
f. lead (II) nitrate, Pb(NO3)2, and barium chloride, BaCl2
4. What is meant by a strong acid? Are the strong acids also strong electrolytes? Explain.