CHEM-UA 126 Chapter Notes -Sodium Acetate, Equivalence Point, Phenol Red
Document Summary
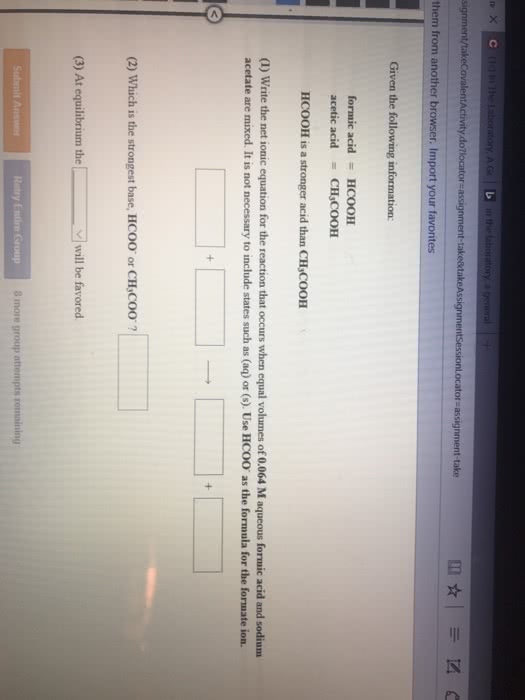
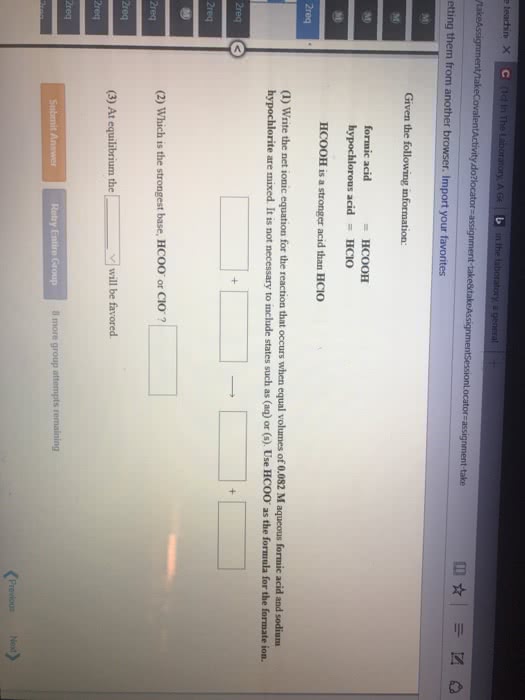
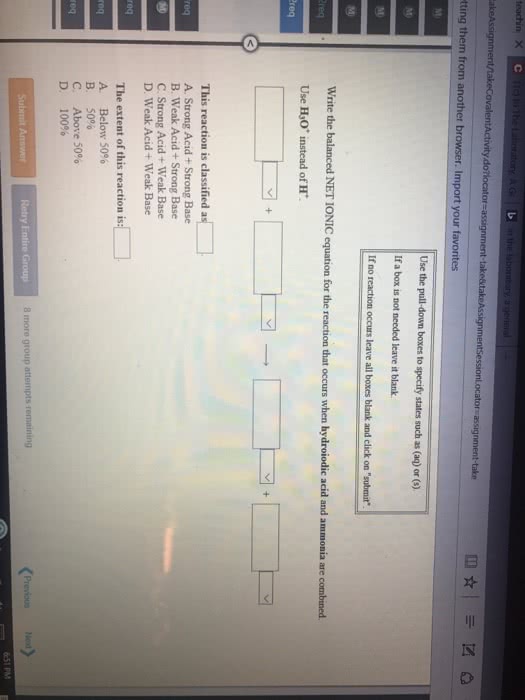
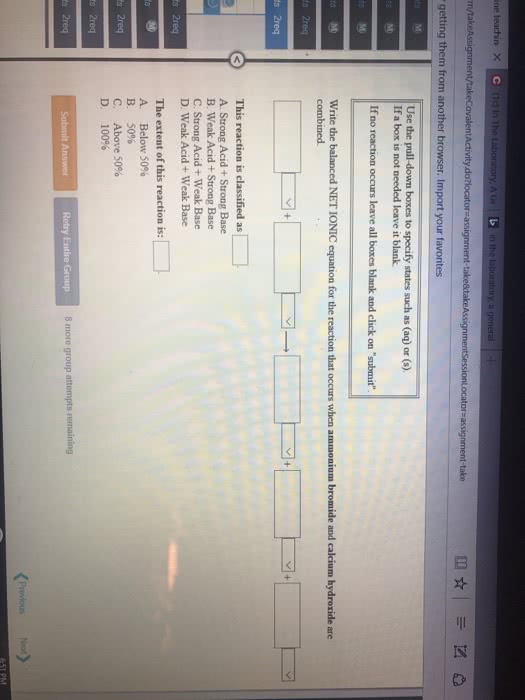
Common ion effect - concerns the position of equilibrium that will be reached when an ion produced by the reaction (i. e. is common to it) is introduced from another source before equilibrium is reached. Adding acetic acid to water will lead to non-negligible concentrations of ch3cooh(aq) . However, if we had included some sodium acetate, there will be another source of acetate ion; this will affect the equilibrium concentrations of all species (in accordance with le chatelier"s principle) In this case, the common ion effect is that ch3cooh is less ionizable; there will be more unionized acetic acid at equilibrium. Ex: compare the ph of a solution that is 0. 30 m in formic acid (hcooh) and 0. 53 m in potassium formate (hcook) to that of a solution that is 0. 30 m formic acid solution; ka = Evaluate the ph of the solution in which the common ion effect is present: Ka = (x*(0. 52 + x))/(0. 30 - x)