CHEM 0110 Chapter Notes - Chapter 9: Electron Affinity, Pauli Exclusion Principle, Electron Configuration
Document Summary
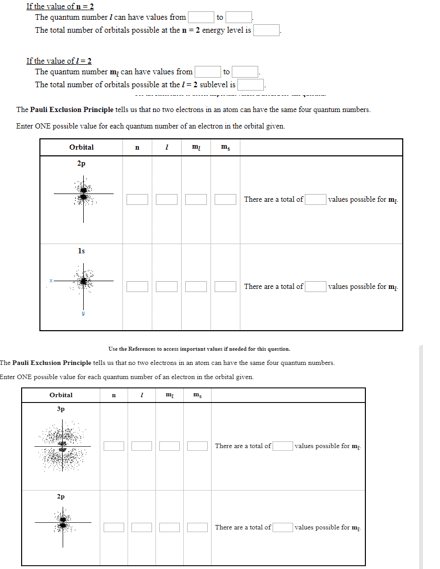
Chem ch 8 notes: electron configuration and periodicity. 8. 1 electron spin and the pauli exclusion principle: When shoot h+ atoms at magnet, the beam split into two, showing that the atoms themselves are magnets. It splits in two b/c the electron in each atom behaves as a magnet with only two possible orientations. Electron spin is subject to quantum restriction on the possible direction of the spin axis. The resulting directions of spin megnetism corresponds to spin quantum numbers. Electron configuration: a particular distribution of electrons among the available subshells. Orbital diagram: shows how the orbitals of a subshell are occupied by electrons. Each orbital is represented by a circle, with two electrons spinning in opposite directions. Pauli exclusion principle: no two electrons in an atom can have the same four quantum numbers. Ie: you cannot place two electrons with the same value of ms in a 1s orbital.