CHEM 227 Chapter 2: Chapter 2 Book Notes
Document Summary
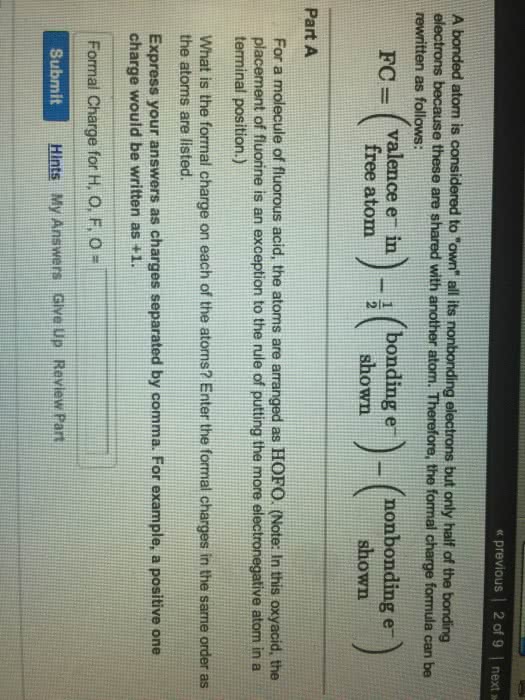
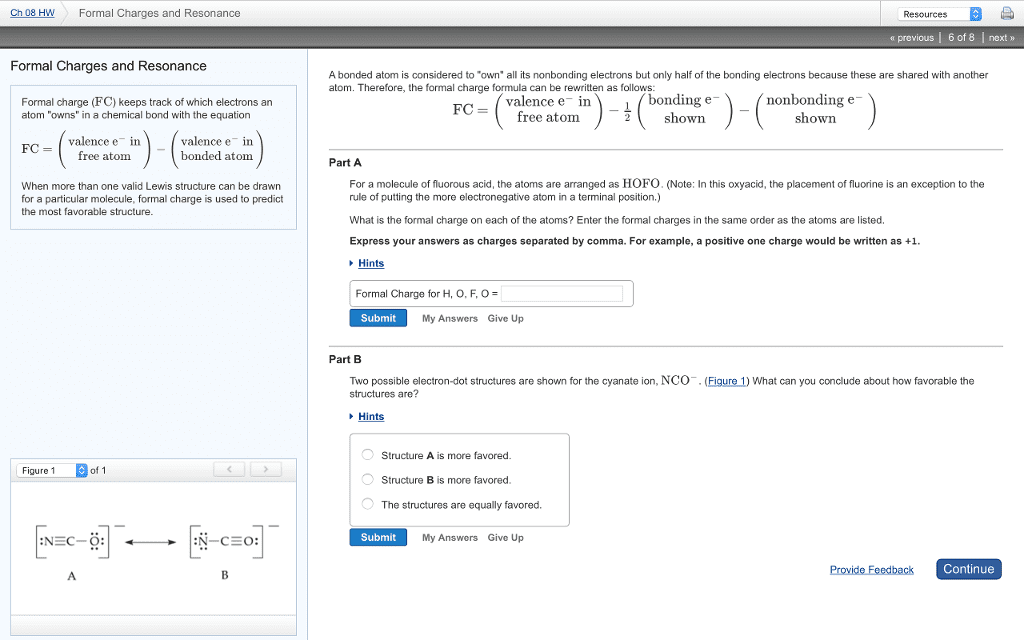
When a bond has an unequal distribution of electrons, one atom will have a partial negative charge while the other has a partial positive charge. This type of bond is known as a polar. The higher the electronegativity the more likely that element will attract electrons. An inductive effect is the shifting of electrons in a sigma bond in response to the electronegativity of nearby atoms. Net molecular polarity is de ned as a dipole moment. Formal charge = (# of valence electrons in free atom) - (# of valence electrons in bonded atom)= (# of valence electrons in free atom) - (# of bonding electrons/2 + number of nonbonding electrons) Formal charge often gives a hint to the reactivity of an atom. The resonance structure of a certain compound is just the different ways certain atoms can be arranged. Even though it will look visibly different the compound will still have the same properties.