CHE 331 Chapter Notes - Chapter 1: Vsepr Theory, Unified Atomic Mass Unit, Electron Configuration
Document Summary
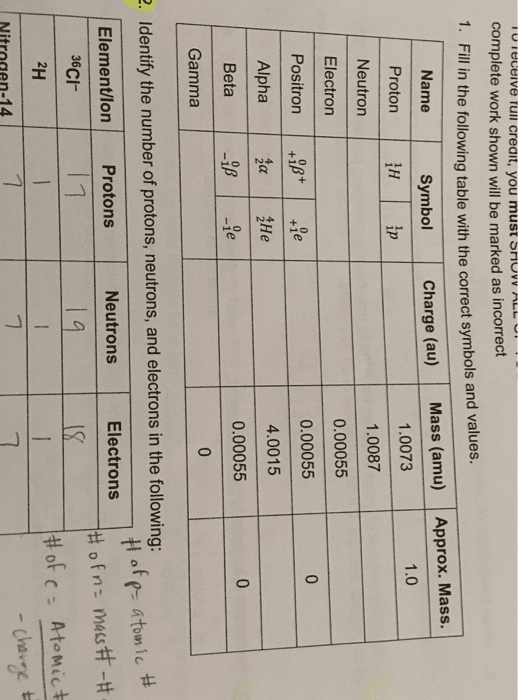
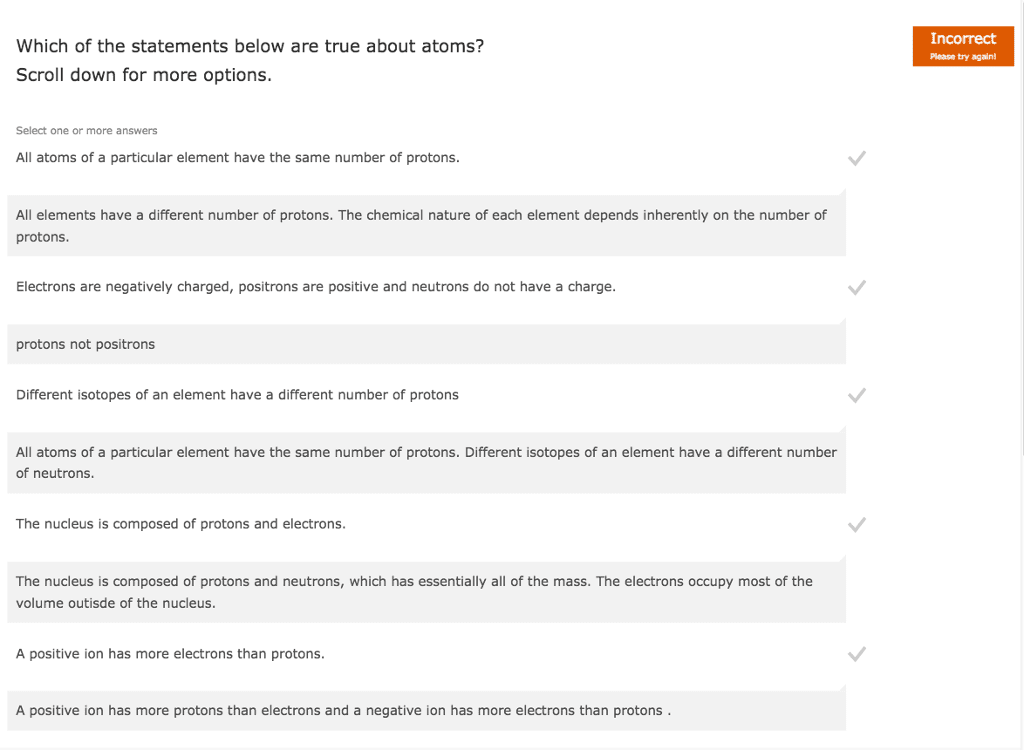
Electrons: most of the volume of an atom comes from the electrons. In nucleus, determines atomic number, name and symbol. Glue of nucleus keeps repelling protons from flying apart. Found outside nucleus in orbitals (regions of space) Isotopes: atoms of the same element with different mass -> different neutron #. Ionic compounds that form when atoms of very different electronegativities transfer electrons: example: sodium chloride (nacl, bond electronegativity difference is 1. 7+ Covalent bonding: co(cid:373)plete t(cid:396)a(cid:374)sfe(cid:396) of ele(cid:272)t(cid:396)o(cid:374)s does(cid:374)"t o(cid:272)(cid:272)u(cid:396) because of similar electronegativities, electron sharing, polar = unequally, non-polar = equally, easily pulled apart when heated, example: water (h2o, bond electronegativity difference is < or = 1. 7, molecules. [# of valence electrons] [electrons in lone pairs (1lp = 2 e-) + 1/2 the number of shared electrons: sum of all formal charges in a molecule or ion will equal the overall charge on it.