CHEM 134 Chapter Notes - Chapter 2: Intermolecular Force, Coordination Complex, Boiling Point
Document Summary
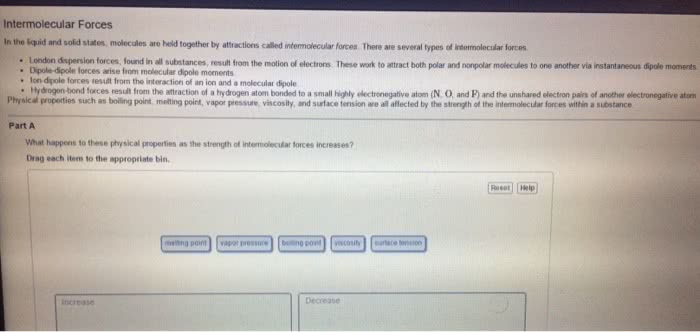
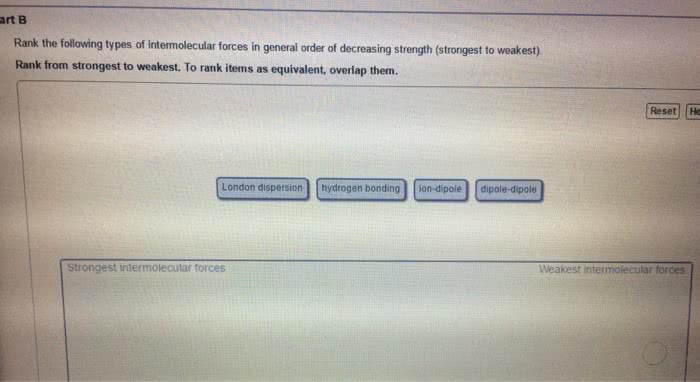
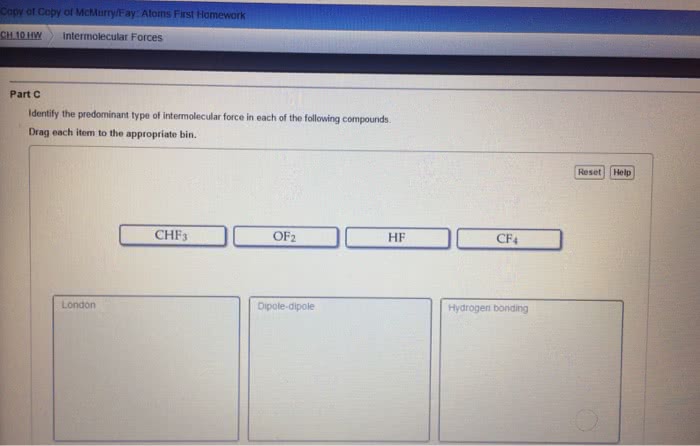
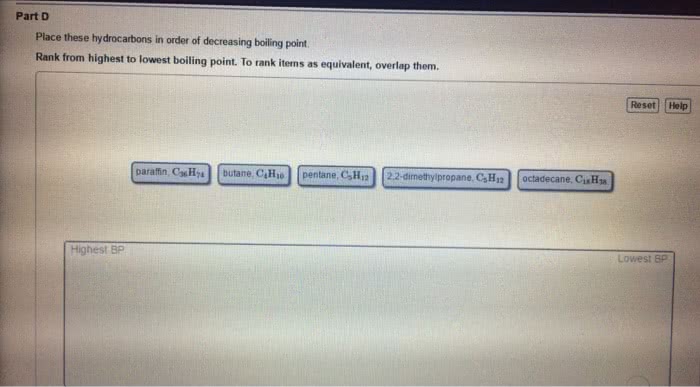
Induced dipole forces are also called dispersion forces, or london forces. H 2 o falls into this category. induced dipole forces. Nicl 2 does not fall into this category. dipole forces. include dipole forces. Hydrogen bonding is found in situations represented by d-h---a where both the donor atom, d , and the acceptor atom, a , are one of the highly electronegative elements o , n , or f . Therefore, ch 3 ch 3 is a nonpolar molecular compound and the only intermolecular forces are induced dipole forces. Therefore, the intermolecular forces also include dipole forces. Hydrogen bonding is not expected to be important in compounds in which all of the h atoms are covalently bonded to c atoms, even when an acceptor atom is present in the molecule. Boiling points increase as intermolecular attractive forces increase. For the alkanes, which are non-polar hydrocarbons, the intermolecular forces are induced dipole forces.