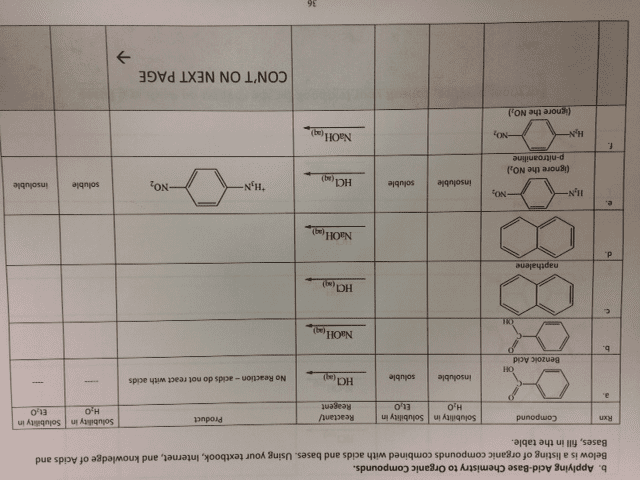
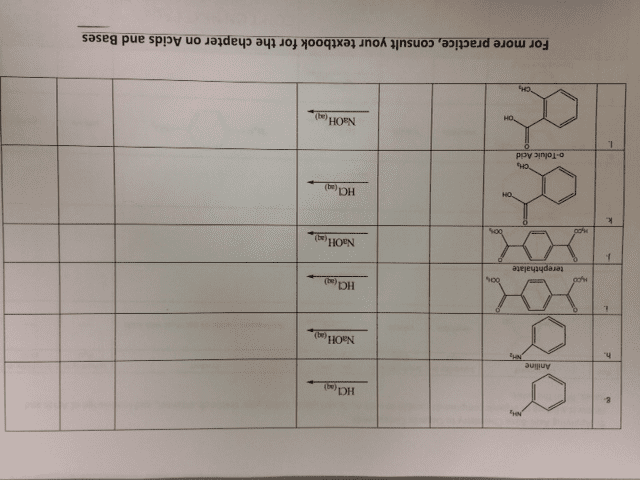
CHEM 1C Chapter 17: Ch 17 Detailed Reading Notes
Document Summary
Bronsted-lowry definition: acids: donate proton (accept e- pair, bases: accept proton (donates e- pair) Strong/weak: a strong acid completely ionizes in solution, whereas a weak acid only partially ionizes, strong base, one that com- pletely dissociates in solution (analogous to a strong acid). A weak base is analogous to a weak acid. Substances that can act as acids or bases are amphoteric. Oxoacids: central element with a bunch of os and hydrogen attached to make oxoacids: when h is removed, structures become resonance structures and oxygen is okay being negative (it"s electronegative). 1st and 2nd groups combined with an oh- group: 1st group wants +1 charge. Naoh, koh, etc: 2nd group wants +2 charge. Nitrogen with three groups/bonds and a lone pair (includes benzene ring with n in it) Hf weak acid: expect f to want neg charge so prefers to be dissociated, bond enthalpy: shorter bonds are stronger.