CHEM 14A Chapter Notes - Chapter 1D.5-6: Samuel Goudsmit, George Uhlenbeck, Magnetic Quantum Number
Document Summary
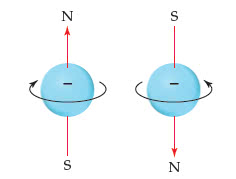
Proposed an explanation for the tiny deviations that had been observed (spectral lines) Suggested that an electron behaves in some respects like a spinning sphere. An electron has two spin states represented by arrows up and down or the greek letters alpha and beta. The two spin states are distinguished by a fourth quantum number, the spin magnetic quantum number (ms) This number has two values: + (up electron) and - (down electron) To summarize, an electron has the property of spin; the spin is described by the quantum number ms = 1/2. In the ground state, the electron is in the lowest energy, the state with n = 1. The electron in the ground state of a hydrogen atom is described by the following values of the four quantum numbers: The electron can have either spin state.