CHEM 1104 Chapter Notes - Chapter 11: Atomic Orbital, Bohr Model, Photon
Document Summary
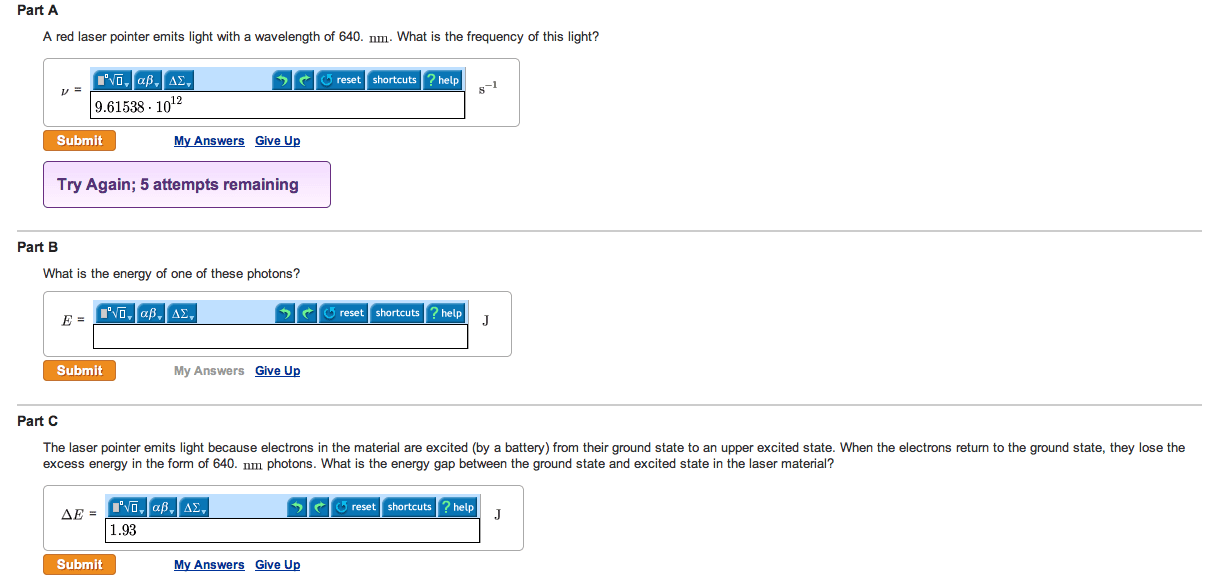
Electromagnetic radiation= energy is being transmitted from one place to another by light. A particular wave is characterized by three properties: wavelength, frequency, and speed. Wavelength= the distance between two consecutive wave peaks. Frequency= indicates how many wave peaks pass a certain point per given time period. Radiation provides an important means of energy transfer. Photons= a stream of tiny packets of energy. The longer the wavelength of light, the lower the energy of its photons. The colors of these flames result from atoms in these solutions releasing energy by emitting visible light of specific wavelengths. An excited atom can release some or all of its excess energy by emitting a photon (a particle or electromagnetic radiation) and thus move to a lower energy state. Ground state= the lowest possible energy state of an atom. Different wavelengths of light carry different amounts of energy per photon.