CHEM 2301 Chapter Notes - Chapter 9: Stereochemistry, Organochloride, Racemic Mixture
Document Summary
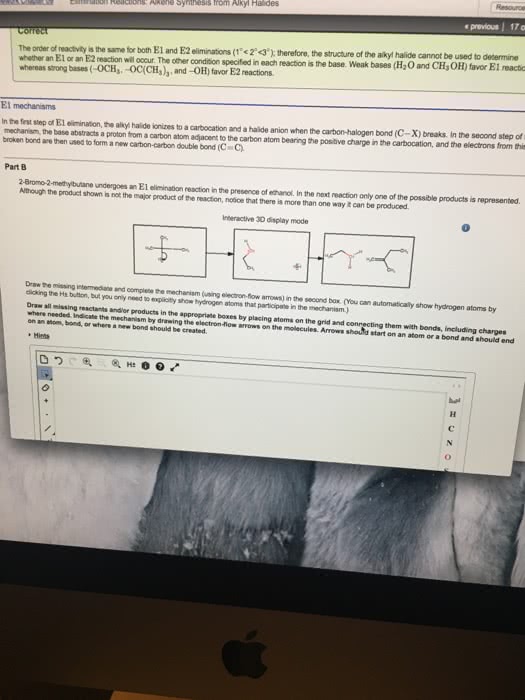
Dehydration = beta elimination reaction in which the elements of oh and h are removed from the alpha and beta c atoms respectively. Typically carried out using h2so4 (& other strong acids) in the presence of an amine base. With different beta c, reaction is regioselective and follows zaitsev rule. More substituted alkene is the major product when a mixture of constitutional isomers is possible. Step 1 - o is protonated (oh becomes h2o) Step 2 - co bond is broken (heterolysis forms a carbocation) Step 3 - base removes a proton from a beta c (new pi bond) Acid used in step 1 is regenerated upon removal of the proton in step 3 - dehydration is acid catalyzed. E1 gives clean elimination products without by products formed from sn1. More synthetically useful than e1 dehydrohalogenation of alkyl halides. Clean because the reaction mixture contains no good nucleophile to react with the intermediate carbocation.