CH 301 Chapter Thermodynamics: Thermodynamics: Long Notes
Document Summary
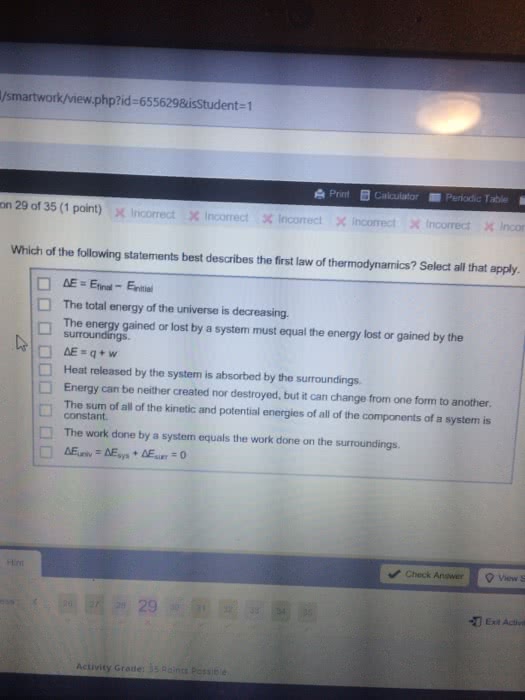
Energy: the ability to do work or produce heat. Heat: a flow of energy due to a temperature difference. Temperature: a measure of the random motions of the components of a substance (average kinetic energy) Law of conservation of energy: energy can be converted from one form to another but can be neither created nor destroyed. Kinetic energy: energy that comes from the motion of the object. Thermal energy: energy that comes from heat. Potential energy: energy that comes from position or composition. Internal energy (u): sum of the kinetic and potential energies of all particles in the system. Chemical energy: the energy stored in the bonds of chemical compounds. Frictional heating: kinetic energy transferred to a surface as heat. Pathway: determines the way an energy transfer is divided between work and heat. State function: property of the system that changes independently of its pathway (can be extensive or intensive) Extensive variables: depend on the amount of material.