CHEM 201 Lecture Notes - Lecture 9: Pauli Exclusion Principle, Atomic Number, Electron Shell
Document Summary
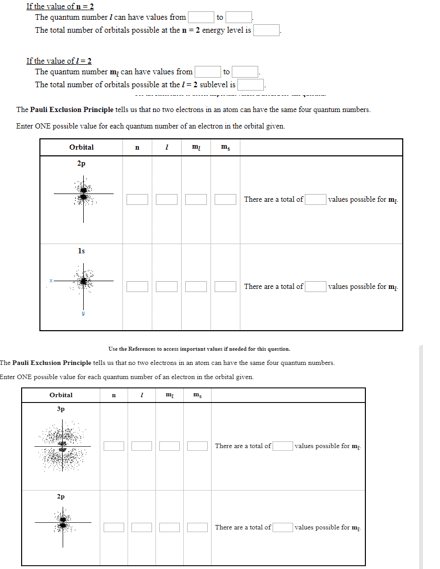
Mendeleev arranged atoms to form the periodic table. He put those that behaved similarly in the same group. He arranged the elements according to the atomic masses. Quantum theory came out with a better way using atomic number. Quantum theory explained that electrons are arranged according to the orbital. One orbital can whole two electrons do opposite spin (m(sub c)) Example: if all boxes of 2p are used it would be 2p^6. Hund"s rule - place one electron to each orbital (if degenerate)before pairing them up. Same electrons are not paired and are affected by a magnetic. Allie"s paired and are not affected by a magnet. Predicting properties of elements - the nucleus more than the outer shell electrons the nucleus to other outer electrons (this is shielding) by the outer shell electrons is called effective nuclear charge. The better the outer electron is at penetrating through the electronic cloud, the more attraction it will have for the nucleus.