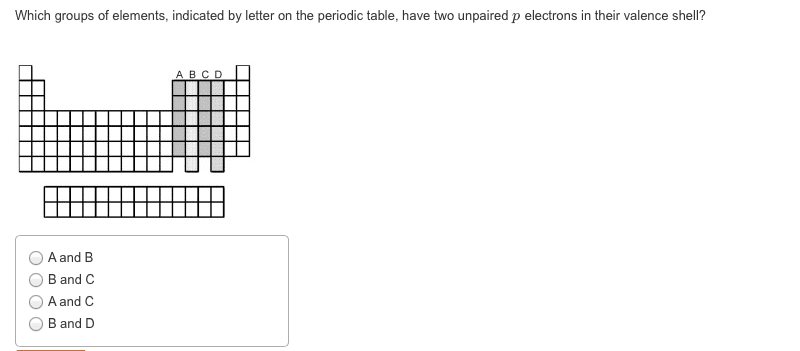CH 101 Lecture Notes - Lecture 4: Radial Distribution Function, Pauli Exclusion Principle, Alkali Metal
Document Summary
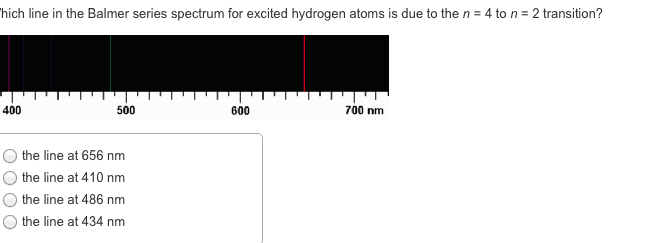
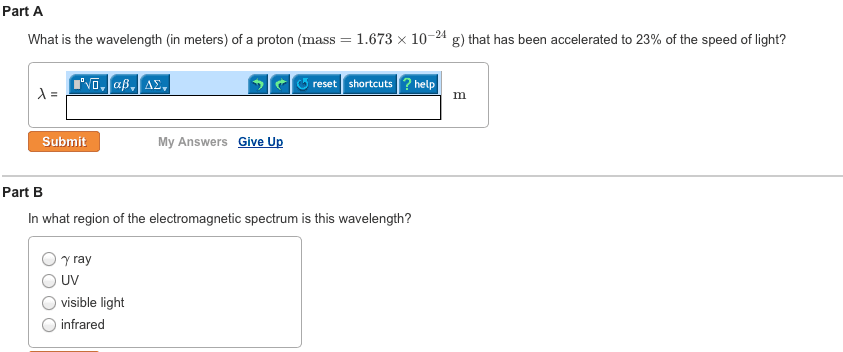
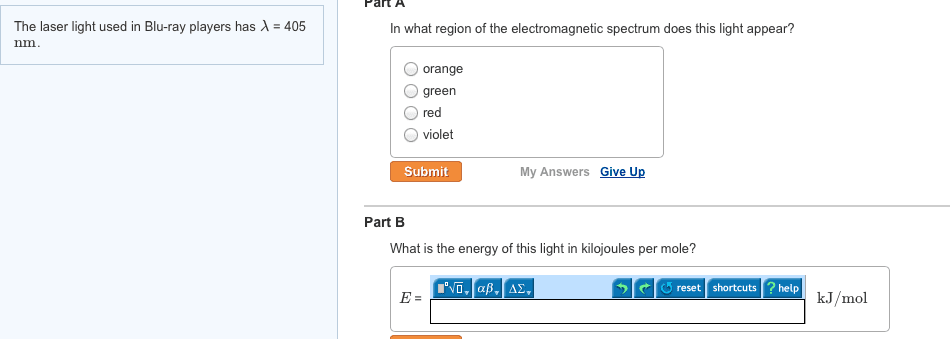
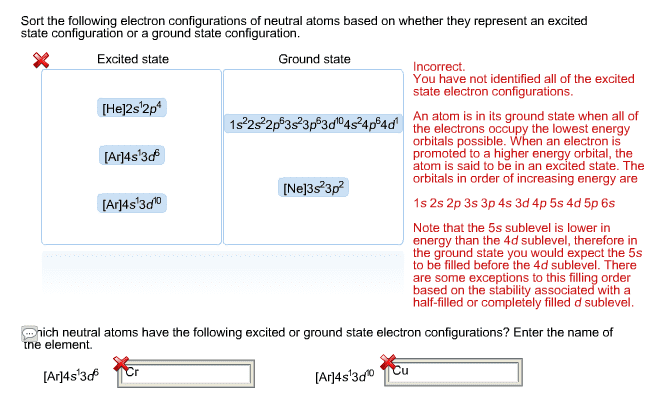
In 1869, mendeleev noticed that certain groups of elements had similar properties. He found that when elements were listed in order of increasing mass, these similar properties recurred in a periodic pattern. When the elements are arranged in order of increasing mass, certain sets of properties recur periodically. Quantum mechanical theory describes the behavior of electrons in atoms. The electrons in atoms exist in orbitals. A description of the orbitals occupied by electrons is called an electron configuration. 1 = number of electrons in orbital. Schrodinger"s equation showed that hydrogen"s one electron occupies the lowest energy level in the atom. For multi-electron atoms, the equation can"t be exactly solved because of the for electron-electron interactions that happen between two electrons. However, approximate solutions showed the orbitals to be hydrogen-like. All electrons have the same amount of spin. It is quantized-can be only in one direction or opposite.