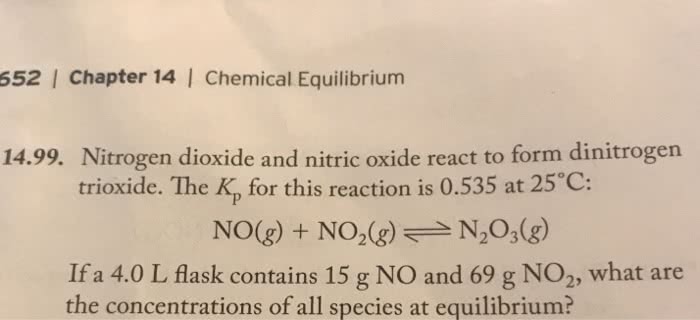CHEN3010 Lecture Notes - Lecture 14: Thermal Conductivity, Computer-Aided Technologies, Heterogeneous Catalysis

Chemical Reaction Engineering
Fogler Chapter 15
Diffusion and Reaction
Semester 1-2018
Gia Hung Pham
find more resources at oneclass.com
find more resources at oneclass.com

Diffusion and reaction
find more resources at oneclass.com
find more resources at oneclass.com

Two steps involve in the reaction (Internal mass transfer and surface
reaction )
Gas
2
4
CAb= CAs
2
4
A
P
Cat-A
Cat-P
Catalyst palette
Catalyst pore
find more resources at oneclass.com
find more resources at oneclass.com
Document Summary
Two steps involve in the reaction (internal mass transfer and surface reaction ) In order to calculate the effective reaction rate we need to know the concentration ca in the catalyst pore. Reaction taking place on the walls of the pore. Material balance for this slice of catalyst pore. First-order reaction is expressed in terms of unit catalyst surface area kv. 2 r r 2 x: divide by. For x (cid:1004) a(cid:374)d i(cid:374) ter(cid:373)s of volu(cid:373)etric u(cid:374)it. This equation can be solved analytically with the boundary conditions (fogler. General solution of the above equation ememc mx. As xlm ml e xlm ml e e. With the boundary conditions: ca = cas at x=0 and dca/dx=0 at x=l. Concentration of reactant a in catalyst pore with m k. Ab and we call ml = 1 (thiele modulus) Single cylindrical pore first order rea(cid:272)tio(cid:374) (cid:894) a p(cid:895) Reaction taking place at the walls of the pore.