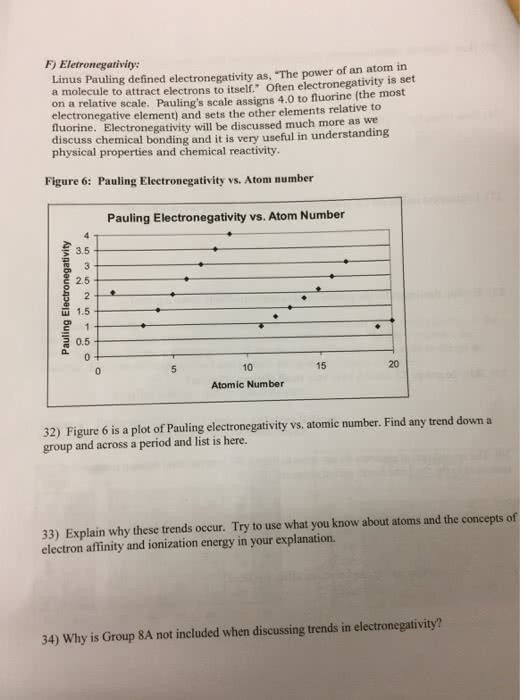
F) Eletronegativity: Linus Pauling defined electronegativity as, The power of an atom in a molecule to attract electrons to itself. Often electronegativity is set on a relative scale. Pauling's scale assigns 4.0 to fluorine (the most electronegative element) and sets the other elements relative to uorine. Electronegativity will be discussed much more as we discuss chemical bonding and it is very useful in understanding physical properties and chemical reactivity. Figure 6: Pauling Electronegativity vs. Atom number Pauling Electronegativity vs. Atom Number 3.5 2.5 1.5 0.5 10 15 20 Atomic Number 32) Figure 6 is a plot of Pauling electronegativity vs, atomie number. Find any trend down a group and across a period and list is here. 33) Explain why these trends occur. Try to use what you know about atoms and the concepts of electron affinity and ionization energy in your explanation. 34) Why is Group 8A not included when discussing trends in electronegativity?