CHEM1831 Lecture Notes - Lecture 9: Chloroform, Ammonia, Intermolecular Force
Document Summary
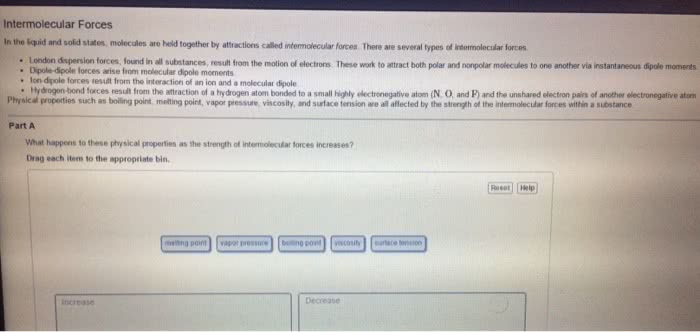
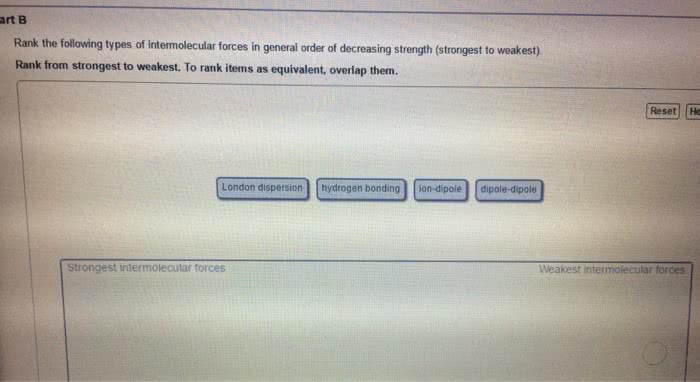
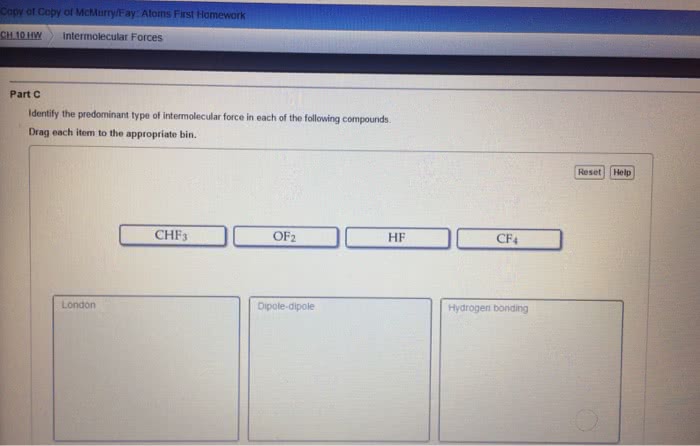
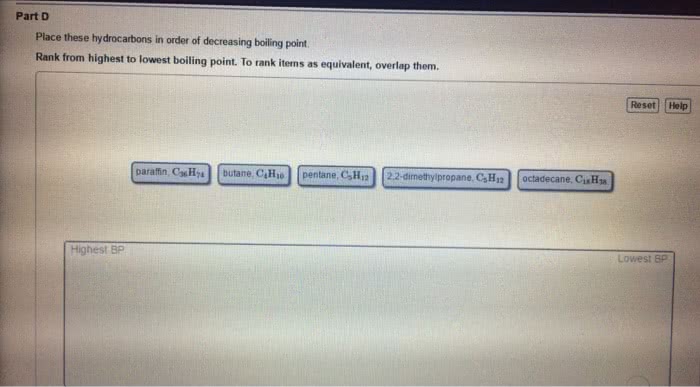
Statins are cholesterol lowering drugs and are divided into those produced naturally by fermentation and those made synthetically. Hydrogen bonds - when a hydrogen atom of one molecule is attracted to a highly electronegative atom e. g f, o, n. Dipole - dipole - attractions between oppositely charged regions of a polar molecular (remember polar bond is one highly electronegative atom bonded to another non electronegative atom) London dispersion forces (van der waals) - induced dipole between nonpolar molecules (note induced forced to be a certain way) typically gases. What holds water molecules together in ice or carbon dioxide molecules in dry ice intermolecular forces. Polar bonds - arise from differences in electronegativities for the bonded atoms. Molecular polarity - is a measure of the asymmetry of the electric charge in a molecule and arises from the individual bond polarities and the overall structure of a molecule.